An Overview Of Virology And Hiv, Including Diagnosis And Treatment Options
INTRODUCTION
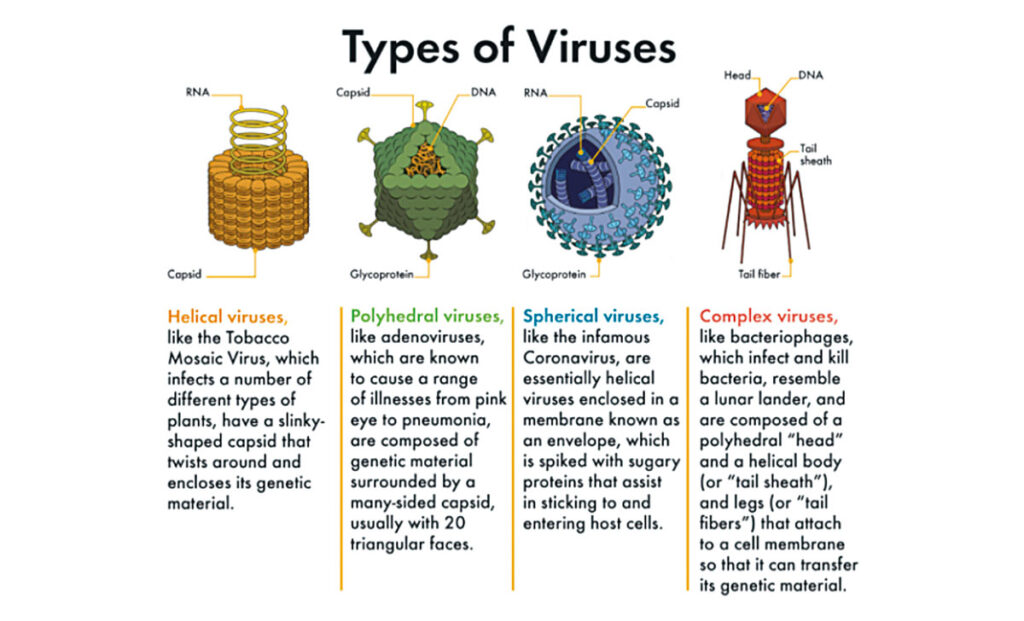
Viruses are submicroscopic, pathogenic microbes that need to use the cellular machinery in other living cells in order to exist.1 Virus genomes are able to direct their own replication using these cellular systems in host cells.2
Because of this inability to reproduce autonomously, technically viruses are not a living organism. However, viruses are still made up of the fabric of all living things, RNA and DNA. At its basic level, a virus consists of a genome with an origin of replication, and a proteinaceous coat (capsid). The two main taxonomic divisions of viruses are those with RNA genomes and those with DNA. Subsequent taxonomic levels are based on the size or structure of the capsid, whether there is a capsid envelope, and the genome structure (single/double stranded, linear/circular). The word ‘virus’ is derived from the Latin word for poison. Infection occurs when viruses, bacteria, or other microbes enter the body and begin to multiply.3
This, however, does not necessarily lead to disease: Disease occurs when cells in the body are damaged as a result of infection, and signs and symptoms of an illness appear. In response to infection, the immune system is activated. Many of the symptoms of an infection (fever, malaise, headache, rash) result from the attempt of the immune system to eliminate the infection. Viruses make us ill by killing cells or disrupting cell function. Our bodies often respond to pathogens with, ie, fever (heat inactivates many viruses), the secretion of interferon (which blocks viral reproduction), or by directing the immune system’s antibodies and other cells to target the invading pathogen.
HUMAN IMMUNODEFICIENCY VIRUS
Human Immunodeficiency virus (HIV) exerts its effects through destruction of CD4 T-cells, eventually causing acquired immunodeficiency syndrome (AIDS). HIV is a retrovirus that replicates in lymphocytes and macrophages, gradually reducing the efficacy of our immune system over time.4
This causes AIDS, which usually develops over 10-to-15 years of HIV infection. AIDS results in a susceptibility to a range of additional opportunistic infections, conditions and malignancies. There are two types of HIV: HIV1, which is responsible for most cases globally; and HIV2, which is mostly in West Africa and is less pathogenic. At the end of 2020, it was estimated that 37.8 million people were living with HIV, with over two-thirds of these people in the WHO African region. Some 36.3 million people have died from AIDS to date; 680,000 people in 2020 alone.5
In Ireland, 449 new HIV cases were diagnosed in 2020. Since the early 1980s, 9,344 people have been diagnosed in Ireland.6 HIV is mostly spread through sexual contact; before birth, during delivery and breastfeeding; and sharing infected needles and syringes.4 For sexually-active adults and people who inject drugs and are knowingly at risk of HIV exposure, pre-exposure prophylaxis to reduce the risk of HIV infection is recommended.
HIV DIAGNOSIS
The symptoms of HIV vary, depending on the stage of infection.5 After initial infection, when people with HIV tend to be the most infectious themselves, common symptoms are swollen lymph nodes, weight loss, fever, diarrhoea, and cough. If left untreated, severe illnesses like tuberculosis, meningitis, severe bacterial infections and cancers can develop. The following diagnostic features are indicative of HIV infection:4 ?
- Fevers and night sweats. ?
- Unexplained weight loss (>10%)/ wasting syndrome. ?
- Skin rashes. ?
- Persistent oral ulcers, angular cheilitis, oral thrush, or oral hairy leukoplakia (caused by Epstein-Barr virus). ? Diarrhoea. ?
- Changes in mental/neuropsychiatric function. ?
- Recent hospital admissions for management of infectious diseases. ?
- Tuberculosis (TB). ?
- Generalised lymphadenopathy (painless enlarged nodes for more than three months). ?
- Kaposi sarcoma (an AIDSdefining condition). ?
- Chronic vaginal candidiasis. ?
- Shingles. ?
- Genital STIs.
Usually, people develop antibodies to HIV within 28 days of infection.5 During these 28 days, people experience a ‘window period’: HIV antibodies are not produced in high enough quantities to be detected, the individual may have no signs of infection, but may transmit HIV to others.
MANAGEMENT OF HIV
Effective antiretroviral treatment can help prevent the development of AIDS by blocking the immune system depletion over time.4 WHO guidelines recommend that as soon as diagnosis is confirmed, all patients with HIV, regardless of CD4 cell count, should start antiretroviral therapy (ART), ideally within seven days of diagnosis or sooner, if there is no other clinical contraindication. ART reduces disease progression, decreases comorbid disease, and prevents transmission of HIV. This recommendation has been reinforced by the findings from the Strategic Timing of AntiRetroviral Treatment (START) study:7 People in the early treatment group showed a 53 per cent reduction in the development of serious illness or death, compared to those in the deferred treatment group. A Cochrane review including 18,000 adults concluded that rapid (within seven days) initiation of ART is linked with greater viral suppression at 12 months.
There are long-term adverse effects of ART, but compared with AIDS itself, these are minimal. Although ART is not a cure for HIV infection, the high rate of viral suppression as a result of administration permits an individual’s immune system to recover. This recovery is sufficient to fight-off opportunistic infections and certain cancers. Globally, 28.2 million of the 37.8 million people living with HIV were receiving ART in 2021.5 In Ireland, HIV care is currently provided in Cork (Cork University Hospital), Dublin (Beaumont Hospital, Mater Misericordiae University Hospital, St James’s Hospital, St Vincent’s University Hospital), Galway (Galway University Hospital), and Limerick (Limerick University Hospital). Paediatric HIV care is provided in Dublin at Temple St University Hospital and Our Lady’s Children’s Hospital. In 2014 and 2016, it was estimated that 92-to-100 per cent of patients here on antiretroviral therapy showed virological suppression rates of >/=90%.8
Highly-active antiretroviral therapy (HAART) refers to when usually three or more antiviral drugs are taken as a combination medication for HIV. As a result of this medication, many people living with HIV have a life expectancy similar to those who do not have HIV.9 In HAART or (‘cART’: Combination antiretroviral therapy) regimens, the multiple drugs that are used inhibit viral replication by several different mechanisms, which means that the propagation of a virus with resistance to one drug is inhibited by the other two drugs.10 There are six different classes of medication used to treat HIV (Table 1). Many treatment-naive patients globally initially receive a combination of two nucleoside reverse transcriptase inhibitors, along with one non-nucleoside reverse transcriptase inhibitor, or integrase strand transfer inhibitor.
The goals of HAART are to: ?
- Reduce morbidity and mortality. ?
- Improve the quality of life. ?
- Reduce plasma viral RNA load. ?
- Prevent transmission to others (sex partners, needle-sharing partners, mother to infant). ?
- Prevent drug resistance. ?
- Improve immune function.
Proper prescribing and adherence to HIV medication is essential for a therapeutic response and prevention of resistance. PREVENTION The risk of HIV infection can be reduced by limiting exposure to risk factors.
Key approaches include:5 ?
- Male and female condom use. ?
- Testing and counselling for HIV and STIs. ?
- Testing and counselling for linkages to TB care. ?
- Voluntary medical male circumcision. ?
- Use of ARVs for prevention. ?
- Harm reduction for people who inject and use drugs. ?
- Elimination of mother-to-child transmission of HIV.
In cases of emergency following possible exposure to HIV via, ie, sex or needle-stick injury, post-exposure prophylaxis (PEP) is available. 11 PEP is a course of medication that can help prevent HIV infection. It is a 28-day course that must be started as soon as possible, but no later than 72 hours, after possible exposure to HIV. PEP is available free of charge in many sexual health/STI clinics around the country.
STEM CELL TRANSPLANTS IN HIV
Three patients with HIV are considered to have been cured. The first, Timothy Ray Brown (known in medical studies as ‘the Berlin patient’), received an allogeneic hematopoietic stem cell transplant (stem cells from another person to replace their own) from a donor with an abnormal CCR5 gene, which confers resistance to HIV. The HIV virus usually uses the CCR5 co-receptor in order to infect immune cells, but this ?32 receptor gene variant effectively blocks viral entry.12 The second was Adam Castillejo, known as ‘the London patient’. Timothy Ray Brown underwent total body irradiation and two adult stem cell transplants from a donor with the CCR5 defect, followed by chemotherapy to kill any residual virus.13 Castillejo underwent similar treatment, with a stem cell transplant from a donor, and chemotherapy, but not total body irradiation. In 2022, reports of the first HIVpositive woman to receive a similar stem cell transplant for acute myeloid leukaemia indicate that she has been in remission and virus-free for 14 months.14 This third patient received transplants of stem cells after having high-dose chemotherapy to destroy her own blood cell population (including leukaemic cells).
The transplanted cells came from two sources: Stem cells from a healthy adult relative, which were used to rapidly restore her blood cell population to reduce complications due to infection; and umbilical cord blood from an unrelated newborn, to provide long-term blood reconstitution. Cord blood containing the HIV-resistance gene variant called CCR5?32 was used.12
It has been suggested that cord blood containing the CCR5?32 variant may offer a possible cure when patients present with both haematological malignancies and HIV, and should be considered as a stem cell source when HIV-positive cancer patients need blood stem cell transplants. The procedure is considered too risky, however, to treat otherwise healthy HIV-positive patients who don’t need such transplants.
Class of Drug | Mechanism of Action | Drug Names |
| Nucleoside/nucleotide reverse transcriptase inhibitors (NRTIs) | Prevention of viral DNA strand growth: These nucleoside analogues with an absent 3’ hydroxyl group that cannot bind to reverse transcriptase and causes premature DNA chain termination | Abacavir, didanosine, lamivudine, stavudine, tenofovir, zidovudine |
| Non-nucleoside reverse transcriptase inhibitors (NNRTIs) | Inhibition of nucleoside binding: NNRTIs bind to HIV reverse transcriptase, causing a stereochemical change that inhibits nucleoside binding and DNA polymerase | Delavirdine, efavirenz, nevirapine, rilpivirine |
| Protease inhibitors (PIs) | Results in the production of immature, non-infectious virions via inhibition of proteolytic cleavage of polyproteins in HIV cells | Atazanavir, darunavir, indinavir |
| Integrase strand transfer inhibitors (INSTIs) | Prevents integration of viral DNA into the host cell chromosome: INSTIs bind to viral integrase | Dolutegravir, elvitegravir, raltegravir |
| Fusion inhibitors (FIs) | Prevent viral fusion to CD4 T-cells | Enfuvirtide |
| Chemokine receptor antagonists (CCR5 antagonists) | Selective and reversible block of HIV entry into CD4 cells | Maraviroc |
References
- Pellett PE, Mitra S, & Holland TC (2014). Basics of virology. Handbook of Clinical Neurology, 123, 45-66. Available https://www.ncbi.nlm.nih.gov/ pmc/articles/PMC7152233/.
- Harvard Museums of Science and Culture (2022). A World of Viruses. Available https://hmsc. harvard.edu/world-viruses.
- Drexler M (2011). What You Need to Know About Infectious Disease: Chapter 1. How Infection Works. Washington (DC): National Academies Press (US). Available https://www. ncbi.nlm.nih.gov/books/NBK209710/.
- BMJ Best Practice (2022). Overview of HIV. Available https://bestpractice.bmj.com/topics/ en-us/1073
- World Health Organisation (2021). HIV/AIDS Factsheet. Available https://www.who.int/newsroom/ fact-sheets/detail/hiv-aids.
- Health Protection Surveillance Centre (2021). HIV Infection in Ireland. Available https://www. hpsc.ie/a-z/hivandaids/.
- Insight Start Study Group. (2015). Initiation of antiretroviral therapy in early asymptomatic HIV infection. New England Journal of Medicine, 373(9), 795-807. Available https://www.nejm.org/ doi/full/10.1056/NEjMoa1506816.
- Health Service Executive (2017). HSE Position on Antiretroviral Therapy for All People Living with HIV. Available https:// www.drugsandalcohol.ie/27848/1/HSE%20 Position%20on%20Antiretroviral%20 Therapy%20FINAL%20090817.pdf.
- Health Service Executive (2017). Antiretroviral Therapy for People Living with HIV in Ireland. Available https://www.hpsc.ie/a-z/hivandaids/ factsheet/ART%20Patient%20Information_ English_vFeb18.pdf.
- Eggleton JS, & Nagalli S (2020). Highly active antiretroviral therapy (HAART). Available https:// europepmc.org/article/nbk/nbk554533.
- HIV Ireland (nd) PEP. Available https://www. hivireland.ie/hiv/prevention/pep/.
- Weill Cornell Medicine (2022). Patient Possibly Cured of HIV Infection by Special Stem-Cell Transplant. Available https://news.weill.cornell. edu/news/2022/02/patient-possibly-cured-ofhiv- infection-by-special-stem-cell-transplant.
- Kuehn Bridget M (2020). Study Suggests a Second Patient Has Been Cured of HIV. JAMA, 323(19), 1886–. doi:10.1001/jama.2020.7626.
- Steenhuysen J (2022). First woman reported cured of HIV after stem cell transplant. Available https://www.reuters.com/business/healthcarepharmaceuticals/ first-woman-reported-curedhiv- after-bone-marrow-transplant-2022-02-15/.