An exploration of the ‘experience’ of pain, including the different types and their characteristics
Nociception is the physiology of actual or potential tissue damage, whereas pain includes the unpleasant thoughts, emotions, and behaviours that accompany nociception.1 It is defined by the International Association for the Study of Pain (IASP) as an unpleasant sensory and emotional experience associated with, or resembling that associated with, actual or potential tissue damage.2 IASP stresses the importance of the fact that pain is always a personal experience that is influenced to varying degrees by biological, psychological, and social factors, such as life experience.
A person’s report of an experience of pain should be respected: Pain intensity can vary widely for a given nociception.
PHYSIOLOGY OF PAIN
The sensation of pain (nociception) is due to the activation of receptors in the primary afferent nerve fibres: The unmyelinated C-fibre and myelinated A?-fibre. Both are silent during homeostasis when there are no nociceptive triggers, and are activated when there is a potential of noxious stimulus.4
In pain perception, a signal is sent via afferent nerve fibres in the peripheral nervous system via the dorsal horn
to higher brain centres (ascending pathway). A response is then propagated to the reflex organs via the spinal cord (descending pathway).
Based on the velocity of the signal conducted and the axon’s diameter and function, the sensory (afferent) neurons are classified into three main groups — Group A, B and C.4
C-fibres is from poorly localised stimuli, ie, burning sensation.
A?- and C-fibres have specialised free nerve endings that are widely located in the skin, muscle, joint capsule, bone, and some major internal organs. They are found in superficial organs, such as the skin, whereas other deep somatic structures, such as muscles and joints, are mainly supplied with C-fibres. Acute inflammatory pain is normally intense and short, and usually mediated by the A?-fibres. However, chronic inflammation lasts beyond the expected period of healing, typically mediated by C-fibres. Silent nociceptors are also present, located within the viscera. These afferent nerve fibres are not activated by physiologically relevant noxious mechanical stimuli, and can only be sensitised by the chemical mediators produced during Studies of musculoskeletal injuries
have found no association between pain intensity and degree of nociception, or injury severity. In fact, variations in pain intensity are accounted for more by measures of psychosocial factors than by measures of pathophysiology. Identifying and addressing psychosocial factors may limit persistent pain.1
Chronic pain lasts beyond the usual course of healing, or more than three-to- six months, which affects the individual’s daily functioning and wellbeing. Several non-modifiable risk factors are linked
to the development of chronic pain: Female sex, age >65 years, intense acute pain, and low socioeconomic status. Several modifiable risk factors have also been identified, including greater pain catastrophising, greater pain- related fear, and greater symptoms of anxiety, depression, and post- traumatic stress disorder.
Type A?: Mainly used to determine proprioceptive function. Group A fibres are all myelinated, meaning they have faster conduction than unmyelinated C nerve fibres.
Type A?: Slow or fast adapting mechanoreceptors.
Type A?: Stretch receptors.
Type A?: Thermal and mechanical nociceptors with relatively fast conduction velocity; responsive to short- lasting and pricking pain.
Group B: These are preganglionic nerve fibres; general visceral afferent fibres belong to this group.
Group C: Unmyelinated, relatively slow conduction velocity; mainly nociceptive
in function (polymodal — activated by thermal, mechanical, chemical stimuli), responsible for about 70 per cent of afferent nociceptive information. The activation of inflammatory reactions. Neuropathic pain is often due to
nerve injury and is often associated with allodynia. It results in a pain response triggered by a stimulus that is usually not painful, ie, pathologic pain. Inflammation or metabolic diseases, such as diabetes, trauma, toxins, tumours, neurological diseases and herpes zoster infection, can all cause neuropathic pain. Neurochemistry of damaged axons can be altered by compression or stretching, which leads to a spontaneous hyper-excitability.
During neuropathic pain, nociceptors demonstrate a dynamic expression of ion channels, ie, sodium channels, which are the major channels involved in neuronal excitability. Neurogenic inflammation occurs when inflammatory mediators, (ie, substance P, calcitonin gene-related peptide, neurokinin) are released locally by the afferent neurons.
Inflammatory pain is a natural biological response to harmful stimuli that helps eradicate the necrotic cells and initiate the tissue repairing process. Neutrophils are usually the first respondents of an inflammatory response and gather at the site of injury via the bloodstream, followed by the release of other chemical mediators. Inflammation can induce mast cell degranulation, leading to the release of platelet-activating factor, causing the release of 5-HT from the circulating platelets. This culminates in a hot inflamed site due to increase in blood flow towards the region, redness, swelling due to vascular permeability, pain caused by the activation and sensitisation of primary afferent neurons, and loss of function.
The localised inflammatory response induces the release of free arachidonic acid from phospholipids, which are converted into prostaglandins (PG) via the cyclooxygenase pathways. Mediators produced at the site of injured tissue (5-HT, kinins, histamine, nerve growth factors (NGF), adenosine triphosphate (ATP), PG, glutamate, leukotrienes, nitric oxide (NO), NE and protons) activate the nociceptors within the inflamed area.
Hyperalgesia refers to a lowered threshold to pain that results in an enhanced perception of pain at the site
of injury. Inflammatory mediators are distributed to receptors at and around the trauma site to cover a larger area than the actual injured region, causing sensitisation of adjacent uninjured tissue to mechanical stimuli (known as secondary hyperalgesia or allodynia). PG is the major component for sensitising procedures of the nociceptors. Primary hyperalgesia has a major peripheral component, while secondary hyperalgesia is due to central sensitisation and mediating mechanisms residing within CNS. Both hyperalgesia and allodynia occur due to the increase of prostaglandin E2 (PGE2) in the inflamed tissue via the activation of COX signalling pathway in the DH of the spinal cord.
The repetitive exposure to noxious stimuli causes a decrease in threshold to stimulation, and increased firing rate due to enhanced sensitivity of primary afferent nociceptors. The released intracellular contents (ATP, bradykinin (BK), 5-HT, NE, PGE2, NGF and SP) at the site of the damaged cells or inflammatory cells, is known as ‘inflammatory soup’. All these inflammatory mediators augment pain sensation through a variety of secondary messengers (ie, cellular cyclic adenosine monophosphate/protein kinase A and protein kinase C/diacylglycerol signalling maintains peripheral hyperalgesia). The end result of peripheral sensitisation is to increase primary afferent nociceptor activity, and pain sensation is enhanced even if the strength of the stimulus remains unchanged.
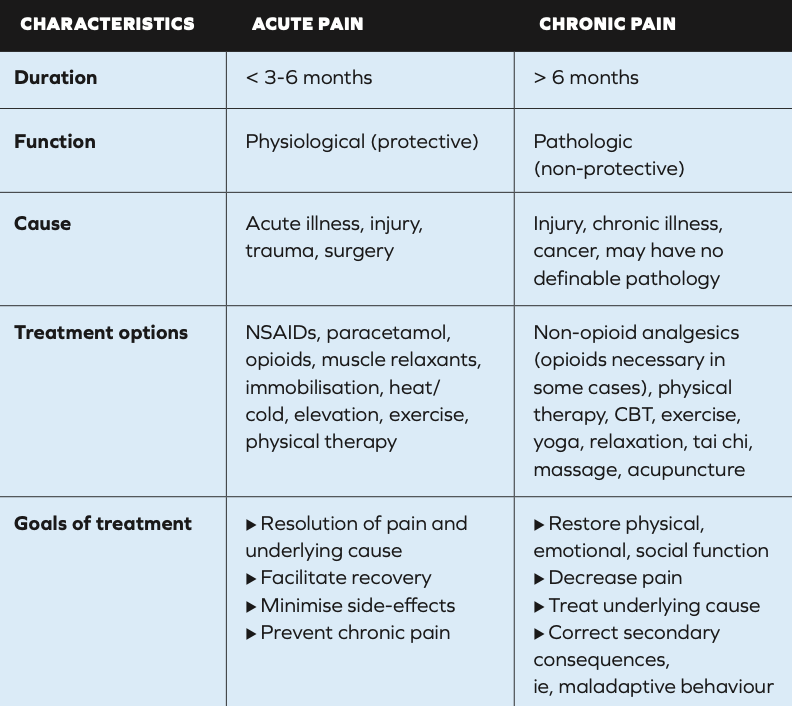
Management considerations based on pain type: Acute vs chronic3
MOST EFFECTIVE OTC ANALGESIA
Authors of a Cochrane Review on the efficacy of OTC analgesics in acute pain5 found that there is evidence to show that simple, inexpensive, readily-available analgesics give good pain relief to many patients with acute pain, such as toothache, sprains and strains. When taking into account the types of OTC products available in Ireland, the most effective OTC drugs for acute pain conditions were ibuprofen/paracetamol combinations in 400mg/1,000mg and 200mg/500mg doses, respectively, providing relief to 70 per cent of people, and with a NNT of less than 2. Fast-acting ibuprofen formulations were effective in over 50 per cent of patients, with an NNT of 2-to-3. Paracetamol helped about 40 per cent of patients and had an NNT of between 3 and 5. Interestingly, the authors identified an absence of evidence to support any efficacy of low-dose codeine preparations in acute pain.
The common advice given with ibuprofen and other NSAIDs to take them with food was also questioned in this review: The absorption of these drugs is faster on having an empty stomach, which increases efficacy. Some ibuprofen product PILs have even been updated to reflect
this advice, ie, the PIL for Nurofen Express Maximum Strength Tablets advises to “take the tablets by mouth only with a glass of water”; no mention of food.6
It was observed that good levels of pain relief were provided by topical NSAIDs in acute conditions such as sprains, strains and overuse injuries, probably similar to that provided by oral NSAIDs.7 A 2020 guideline8 developed by the American College of Physicians (ACP) and American Academy of Family Physicians (AAFP) for management of acute pain from non–low back, musculoskeletal injuries in adults is in agreement: Topical NSAIDs were among the most effective interventions. Furthermore, topical NSAIDs were not associated with a statistically significant increase in risk for adverse effects.
Topical diclofenac, ibuprofen, and ketoprofen in particular demonstrated significantly higher rates of clinical success (more participants with at least 50 per cent pain relief) than matching topical placebo. For diclofenac, the Emulgel formulation had the lowest NNT of 1.8 for at least 50 per cent pain intensity reduction. Diclofenac plasters also had a low NNT of 3.2. Ibuprofen gel had an NNT of 3.9 as measured by outcomes of marked improvement or complete remission.7 The ACP and AAFP guidelines state that oral NSAIDs and paracetamol both reduce pain, however, oral NSAIDs were associated with increased risk for GI adverse events.8
NON-PHARMACOLOGICAL PAIN INTERVENTIONS
Some trials indicate that cognitive behavioural therapy (CBT) interventions have positive effects on pain relief, including an online CBT intervention. Meta-analyses of music therapy show a reduction in anxiety levels and better sleep in patients with chronic illness.1 Music therapy has also shown positive effects on pain relief and opioid dose reduction. Similarly, systematic reviews of aromatherapy have demonstrated anxiolytic effects and pain reduction. TENS has also been found to be an effective multimodal adjunct to modulate acute orthopaedic injury and postoperative pain.
There appears to be some support for use of cryotherapy and heat therapy (particularly for lower back pain) for acute pain management.9 However, the most effective method of application and protocol for these still needs to be determined.
Where necessary, the use of multiple analgesic medications in conjunction with non-pharmacologic interventions can be very effective for greater pain control.1 This is due to the potentiation of multiple medication effects without relying solely on any one class of medication, potentially mitigating the risk profile of each medication, while allowing for synergistic pain control from different drug classes.
Author: Dr Donna Cosgrove PhD. Donna’s overall aim is to improve patient outcomes through education. After graduating with a BSc in Pharmacy, she returned to university to complete a MSc in Neuropharmacology. This led to a PhD investigating the genetics of schizophrenia, followed by a postdoctoral research position in the same area. She has worked in hospital, research and community pharmacy settings and is currently a community pharmacist in Galway and a clinical writer.
REFERENCES
1. Hsu JR, Mir H, Wally MK, & Seymour RB (2019). Clinical practice guidelines for pain management
in acute musculoskeletal injury. Journal of Orthopaedic Trauma, 33(5), e158. Available https://www. ncbi.nlm.nih.gov/pmc/articles/ PMC6485308/#R15.
2. International Association for the Study of Pain (2021). IASP announces Revised Definition of Pain. Available https://www.iasp-pain. org/publications/iasp-news/iasp- announces-revised-definition-of-pain/.
3. American Academy of Family Physicians. (2022). Chronic Pain Toolkit. Available https://www.aafp.org/dam/ AAFP/documents/patient_care/pain_ management/cpm-toolkit.pdf.
4. Yam MF, Loh YC, Tan CS, Khadijah Adam S, Abdul Manan N, & Basir R (2018). General pathways of pain sensation and the major neurotransmitters involved in pain regulation. International Journal of Molecular Sciences, 19(8), 2164.
5. Moore RA, Wiffen PJ, Derry S, Maguire T, Roy YM, & Tyrrell L (2015). Non-prescription (OTC) oral analgesics for acute pain – an overview of Cochrane reviews. Cochrane Database of Systematic Reviews (11).
6. Reckitt Benckiser Healthcare International Ltd (2020). Nurofen Express Maximum Strength 400mg Tablets Patient Information Leaflet. Available https://www.medicines.ie/ medicines/nurofen-express-maximum- strength-400mg-tablets-33157/ patient-info.
7. Derry S, Moore RA, Gaskell H, McIntyre M, Wiffen PJ. Topical NSAIDs for acute musculoskeletal pain in adults. Cochrane Database of Systematic Reviews 2015, Issue 6. Art. No.: CD007402. DOI: 10.1002/14651858.CD007402.pub3. 8. Qaseem A, McLean RM, O’GurekD, Batur P, Lin K, Kansagara DL, & Clinical Guidelines Committee of the American College of Physicians and the Commission on Health of thePublic and Science of the American Academy of Family Physicians* (2020). Nonpharmacologic and pharmacologic management of acute pain from non–low back, musculoskeletal injuries in adults: A clinical guideline from
the American College of Physicians and American Academy of Family Physicians. Annals of Internal Medicine, 173(9), 739-748.
9. Chou R, Wagner J, Ahmed AY, Blazina I, Brodt E, Buckley DI, … & Skelly AC (2021). Treatments for acute pain: A systematic review. Available https://europepmc.org/article/nbk/ nbk566506.