This module focuses on the clinical management of rheumatoid arthritis (RA) and psoriatic arthritis (PsA). On completion of this module, it is expected the reader will have an enhanced understanding of the diagnostic and treatment considerations, including follow-up and monitoring, and tapering or discontinuation of medicines.
Diagnosis and early recognition of RA and PsA RA and PsA are chronic inflammatory arthritic diseases that cause joint pain, swelling, stiffness, and progressive joint damage. RA is an autoimmune condition primarily affecting synovial joints symmetrically, with a prevalence of about 1 per cent in Western populations. PsA is an inflammatory arthritis associated with psoriasis and it can involve peripheral joints.
Both conditions can lead to significant disability and systemic complications if not treated early and aggressively.
Treatment involves a combination of symptomatic therapies — analgesics, nonsteroidal anti-inflammatory drugs (NSAIDs), and corticosteroids — and disease-modifying antirheumatic drugs (DMARDs) to slow disease progression.
These include conventional synthetic DMARDs (cedars) like methotrexate; newer biological DMARDs (bDMARDs) such as tumour necrosis factors (TNF) inhibitors; and targeted synthetic DMARDs (tsDMARDs) like Janus kinase (JAK) inhibitors.
Timely diagnosis of RA is critical as early intervention improves long- term outcomes. NICE Guideline NG100 (2018) underscores a referral to a rheumatologist for any adult with suspected persistent synovitis of unknown cause. Certain features heighten suspicion of RA and warrant urgent referral, including involvement of small joints of the hands/feet, multiple joint involvement, or symptom duration of greater than three months.
Even if rheumatoid factor or anti-CCP antibody tests are negative, referral should be based on clinical examination since seronegative RA can occur. Early rheumatology evaluation — ideally within three months of symptom onset — allows prompt initiation of DMARD therapy, which can achieve remission and prevent irreversible joint damage. At diagnosis, baseline disease activity and prognostic factors — eg, high acute phase reactants, positive anti-CCP, early erosions on x-ray — are assessed, as these indicators of poor prognosis call for more aggressive therapy.
Monitoring and follow-up
The goal in RA is to achieve absence of active inflammation or low disease activity if remission is not attainable. Disease activity is measured by composite scores such as DAS28, which incorporates swollen/tender joint counts and inflammatory markers (CRP/ ESR). Remission is typically DAS28 <2.6, and low disease activity ?3.2.
The DAS score was initially developed in the Netherlands in the early 1990s using a combination of diagnostic criteria including symptoms — eg, swell points — and RA indicators in blood tests to allow clinicians to determine RA severity. The DAS score has been modified to simplify diagnosis since it was developed — eg, reducing number of symptoms a clinician must look for — to become an internationally accepted measurement tool to help diagnose and indicate RA severity.
Patients with active RA should have their therapy escalated until the target is reached. According to NICE NG100, once in remission or low disease activity, patients should be monitored for at least six months to ensure stability. Early and sustained control of inflammation though treatment
has been shown to improve long-term functional and radiographic outcomes in RA.
Treatment options
What are DMARDs?
As DMARDs are standard pharmaceuticals for treatment of both RA and PSA, I want to start with a summary of DMARDs as they will be referred to a lot in this article. DMARDs stands for disease-modifying anti- rheumatic drugs.
DMARDs are a class of medications used to treat autoimmune conditions such as:
- Rheumatoid arthritis;
- Psoriatic arthritis;
- Ankylosing spondylitis; and
- Systemic lupus erythematosus (SLE).
Unlike symptomatic treatments such as NSAIDs or corticosteroids, DMARDs aim to alter the course of the disease by suppressing the underlying immune response that causes inflammation and joint damage.
Brief summary of types of DMARDs
Conventional DMARDs — for example, methotrexate, leflunomide — work by broadly suppressing the immune system to reduce inflammation and slow joint damage in RA. They affect multiple immune pathways and may take weeks or months to show benefit. Biological DMARDs are newer, targeted therapies, often monoclonal antibodies, which block specific immune signals like TNF-?, IL-6, or B-cells. Biologics usually act faster and more precisely but are more expensive and carry a higher risk of infections.
Rheumatoid arthritis
Start DMARDs as soon as possible to limit joint damage To treat RA, DMARDs should be started as soon as possible after diagnosis. Current guidelines recommend this — usually methotrexate, leflunomide, or sulfasalazine — as first-line, rather than immediate combination therapy.
This approach marks a change from the older practice of starting multiple DMARDs together; however, recent evidence did not demonstrate combination therapy to be superior to a step-up approach. Methotrexate is often the anchor drug due to its efficacy in symptom control and radiographic progression slowing. It can be used alone or in combination with other DMARDs and is typically titrated up to 15-25mg weekly as tolerated. Folic acid supplementation is given to reduce methotrexate side-effects.
If the initial DMARD monotherapy at an adequate dose for approximately three months does not achieve the target, the options are switching to an alternative conventional DMARD (csDMARD) or adding a second conventional DMARD (step-up combination). Combination csDMARD therapy — eg, methotrexate + sulfasalazine ± hydroxychloroquine, known as ‘triple therapy’ — is also an option, though in practice many patients move to biologic therapy if two conventional agents fail to control moderate-to-severe disease.
Symptomatic control until the benefits of DMARDs take effect
Because csDMARDs have a slow onset (often six-to-12 weeks for effect), patients with active RA
often require interim symptom relief. Simple analgesics like paracetamol are commonly used to help control pain. Notably, NICE found limited evidence for use of paracetamol or weak opioids in RA, and the latest guidance emphasises NSAIDs as the preferred analgesic for inflammatory pain. However, paracetamol can be used adjunctively and regular dosing of paracetamol may enhance NSAID pain relief and allow lower NSAID doses. Opioid analgesics — eg, codeine or tramadol — are reserved for short-term use during severe flares or if NSAIDs are contraindicated. Long-term opioid use is discouraged due to risks of dependence, sedation, constipation, and falls in the elderly.
More on symptomatic relief
NSAIDs reduce inflammation, pain, and stiffness, providing symptomatic benefit in RA. They are commonly used to manage acute joint symptoms. For example, ibuprofen, naproxen, or COX- 2 selective inhibitors like etoricoxib may be used. NSAIDs can significantly improve quality of life in acute RA,
but their well-known toxicities require caution. Gastrointestinal ulceration/bleeding, renal impairment, hypertension, and cardiovascular risk — especially with diclofenac or
COX-2 inhibitors — are important considerations. NICE NG100 advises co-prescribing a proton pump inhibitor (PPI) for gastric protection when using NSAIDs, and regular review of NSAID need and patient risk factors. Only one NSAID should be used at a time and long-term continuous use is avoided, if possible, particularly in patients with cardiovascular or renal comorbidities.
Glucocorticoids have potent anti- inflammatory effects and can serve as ‘bridge therapy’ in RA. Low-dose oral prednisone or intramuscular methylprednisolone can provide rapid symptom relief while awaiting DMARD efficacy. Corticosteroids also have some disease-modifying properties in RA — they can slow joint erosion progression — but due to their long- term side-effects, NICE encourages rheumatologists to use short-term for symptom control when starting a new DMARD. In practice, a patient might receive a six-to-12 week tapering course of oral prednisolone or periodic depot steroid injections early in treatment. Intra-articular steroid injections are very useful for localised inflammation; for example, a swollen knee can be injected with methylprednisolone acetate (Depo-Medrone), leading to rapid relief without systemic side- effects. Chronic or frequent steroid use is avoided due to significant adverse effects: Osteoporosis, weight gain, hyperglycaemia, hypertension, skin thinning, and infection risk. Current practice is to taper and discontinue corticosteroids ideally within six months of achieving disease control.
Conventional DMARDs (csDMARDs)
Methotrexate (MTX) is considered a first-line csDMARD for most RA patients due to its efficacy and long- term tolerability. It can be given orally or subcutaneously once weekly. MTX typically starts at 10-to-15mg weekly and can be escalated (up to 20-to- 25mg weekly) as tolerated. The onset of benefit is in six-to-12 weeks, so patients should be counselled to continue the medication even if they do not feel immediate improvement. MTX is often combined with other therapies — it synergises with biologics and can reduce anti-drug antibody formation against biologic agents.
Common side-effects include gastrointestinal upset, oral ulcers, alopecia, and fatigue. More serious risks are hepatotoxicity, bone marrow suppression, and pneumonitis. Testing is therefore mandatory — typically full blood count and liver enzymes every two-to-four weeks at therapy initiation, then spacing out to every eight-to-12 weeks once stable. Folic acid 5mg weekly (on a different day than MTX) is given to mitigate mucosal and hepatic side-effects.
Pharmacists dispense MTX with a clear dosing schedule to avoid the grave error of daily dosing and educate patients on signs of toxicity (ie, mouth sores, infection, cough, or shortness of breath). Women of childbearing potential must be counselled on strict contraception during and after MTX, as it is teratogenic, and to stop MTX and consult their doctor if planning pregnancy.
Sulfasalazine is an anti-inflammatory and immunomodulatory agent that is often used as first-line or in combination with MTX. It is taken orally (gradually titrated to 2-3g/ day in divided doses) and may take one-to-three months for effect. Sulfasalazine is safe, though it can cause gastrointestinal upset (nausea, dyspepsia) and reversible sperm count reduction in men. It requires monitoring of blood counts and liver function tests (LFTs) as well. It has shown similar
Pharmacists should advise that enteric- coated tablets help tolerance
one-year efficacy to MTX in some studies. Pharmacists should advise that enteric-coated tablets help tolerance, and that sulfasalazine can cause orange discoloration of urine or tears — harmless, but useful for contact lens users to know.
Leflunomide is a potent immunosuppressant (pyrimidine synthesis inhibitor) and is used especially if MTX is not tolerated. It is given as a daily oral dose (10-to- 20mg). Leflunomide is effective but
can cause liver enzyme elevations, hypertension, and is highly teratogenic. Its long half-life necessitates a cholestyramine washout if urgent drug removal is needed; for example, in case of pregnancy. Monitoring is similar, requiring LFT and complete blood count.
Hydroxychloroquine is an antimalarial with mild DMARD effect, often used in mild RA or as part of combination, eg, MTX-sulfasalazine- hydroxychloroquine triple therapy. It is well tolerated and does not require blood test monitoring for toxicity. However, hydroxychloroquine can cause retinal toxicity with long-term use; patients need baseline and yearly ophthalmologic exams. Pharmacists should remind patients to receive eye checks and report any visual changes. Hydroxychloroquine is one DMARD that is safe in pregnancy if needed.
Azathioprine and cyclosporine are sometimes used in refractory RA — ie, RA which is not responding to other treatment options — or specific scenarios, for example, cyclosporine may be used for RA with vasculitis overlap, a rare situation where RA is also associated with inflammation of blood cells.
Azathioprine and cyclosporine are rarely considered due to their higher toxicity profile. Azathioprine can cause cytopenias and liver toxicity (TPMT enzyme activity may be checked before use), and cyclosporine can cause nephrotoxicity and hypertension. These require specialist use and close monitoring.
Biologics
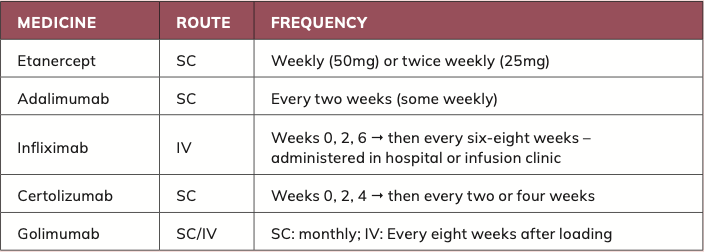
Biologics can be used if a patient (by NICE definition, eg, DAS28 >5.1) is inadequately controlled with conventional DMARD. In Ireland and the UK, typically the patient must have tried at least two csDMARDs (including MTX) and still have active disease to access biologics under reimbursement criteria. Biologics are targeted therapies derived from biologic processes that block specific inflammatory cytokines or cells. Major classes and agents include:
- Etanercept (Enbrel and biosimilars);
- Adalimumab (Humira and biosimilars);
- Infliximab (Remicade and biosimilars) – administered in hospital or infusion clinic;
- Certolizumab (Cimzia); and
- Golimumab (Simponi).
These were the first biologics approved for RA and remain widely used. They neutralise TNF, a key pro-inflammatory cytokine in RA pathogenesis. In Ireland, etanercept and adalimumab are among the most-prescribed biologics for RA. These agents can produce dramatic improvements in symptoms and function, often within one-to-two months. They are typically given by subcutaneous injections — weekly or biweekly for etanercept, biweekly for adalimumab — except infliximab, which is an IV infusion. All TNF inhibitors are often used in combination with methotrexate, which improves efficacy and reduces anti-drug antibody formation, though they can be used as monotherapy if MTX is contraindicated. Patients and pharmacists should know the proper storage — refrigeration — and handling of injectable biologics. Infections and headaches are common side-effects. Serious risks include reactivation of latent tuberculosis or hepatitis B and increased susceptibility to serious infections. Therefore, prior to starting any biologic, patients undergo screening for TB (TB skin or IGRA test, chest x-ray) and hepatitis. They also should be up-to-date with immunisations (influenza, pneumococcal, etc). Live vaccines are contraindicated while on biologics.
Pharmacists should counsel patients to promptly report fevers or signs of infection. Because TNF blockers suppress the immune response, rarer risks like lymphoma or other malignancies have been observed. Although absolute risk is low, patients should undergo regular skin checks for melanoma and be vigilant about cancer screening.
Other biological DMARD options
Tocilizumab (IV or SC) is a monoclonal antibody against IL-6 receptor, effective in RA especially if TNF inhibitors fail. It can cause elevated cholesterol and abnormal LFTs, so monitoring is needed.
Rituximab (an anti-CD20 monoclonal antibody) selectively depletes B lymphocytes. It is given as two IV infusions about two weeks apart and can be repeated every six-to-12 months as needed. Rituximab is often used in seropositive RA or when TNF inhibitors are ineffective. It carries infusion reaction risks and requires premedication. It also can reactivate hepatitis B (hence screening is required).
Abatacept is a fusion protein that inhibits T-cell activation. It is another option for moderate-to-severe RA, given IV or SC.
Biosimilars
In recent years, versions of biologic drugs — biosimilar of infliximab, etanercept, adalimumab — have become available in Ireland. These have comparable therapeutic effects and safety to the reference biologics and are less expensive, thereby offering cost savings. The HSE has promoted switching stable patients to biosimilars to reduce costs. Community pharmacists currently cannot substitute biologics without prescriber approval (unlike generic small-molecule drugs), but the Irish health system has implemented policies to encourage biosimilar uptake. The Irish Pharmacy Union has even proposed that pharmacists be empowered to dispense a biosimilar in place of a biologic on first issuance, to streamline adoption.
From a pharmacist perspective, being knowledgeable about biosimilars and reassuring patients of their equal efficacy is important, as some patients express anxiety about switching from a familiar biologic to a biosimilar. Clear communication that biosimilars are rigorously assessed and approved by the European Medicines Agency for equivalence can improve acceptance. This is one area where pharmacists significantly contribute to supporting biosimilar use.
JAK inhibitors
The newest category of DMARDs are oral small molecules that inhibit Janus kinase enzymes involved in cytokine signaling.
They are indicated for moderate-to- severe RA after inadequate response or intolerance to conventional DMARDs — they can be used either with MTX or as monotherapy. JAK inhibitors have expanded treatment options, especially for patients who prefer an oral agent over injections. Studies demonstrate that many RA patients appreciate the oral route and quicker onset of action of JAK inhibitors. Indeed, JAK inhibitors may begin improving symptoms within one-to-two weeks in some cases, whereas biologics can take multiple weeks. However, because their mechanism broadly affects immune signaling, JAK inhibitors carry similar infection risks as biologics and some unique concerns. Notably, tofacitinib and baricitinib have been linked to increased herpes zoster (shingles) reactivation — vaccination for shingles, with the non-live Shingrix vaccine, may be considered in eligible patients.
Other risks include elevated cholesterol and rare events like venous thromboembolism, particularly at higher doses or in high-risk patients. In 2021, safety warnings highlighted a possible increased risk of major cardiac events and malignancy with JAK inhibitors in certain populations, prompting more cautious use, eg, lowest effective dose and avoiding those with past malignancy if possible. As a result, regulatory agencies advise that JAK inhibitors be used only after at least one biologic DMARD has failed, in patients over 65, or with risk factors, due to these safety signals. Checks for TB, hepatitis, etc, are done before initiation, just as with biologics.
Live vaccines are contraindicated while on JAK inhibitors. Regular blood monitoring is required (for cytopenias, liver enzymes, lipids). Patients should be counselled on promptly reporting infections, paying attention to symptoms such as fever, cough, etc, or reporting thrombotic symptoms — leg pain/swelling, shortness of breath. It is important to avoid sudden discontinuation without medical advice. JAK inhibitors, having short half-lives, may allow quicker reversal of adverse effects upon stopping compared to biologics.
Tapering or discontinuing
A subset of RA patients will achieve sustained remission on combination therapy. At that stage, a common question is whether medications can be tapered or stopped. Guidelines suggest cautious discontinuations may be considered in patients who have maintained remission for at least six- to-12 months.
The 2018 NICE RA guideline (Guidance NG100) recommends rheumatologists consider a step-
down reduction of drugs after one year of sustained remissions. EULAR (European League against Rheumatism) guidance (2016) also supports the consideration of tapering, particularly of biologics, after sustained remission, but emphasises close monitoring if remission persists.
Any attempt should try to gradually taper biologic DMARDs, eg, lengthening dosing intervals rather than abrupt cessation. This is because sudden withdrawal of biologics can lead to high flare rates. Finally, if the patient remains in drug-free remission, one might cautiously taper the csDMARD (eg, MTX) — though in practice all RA therapy often results in disease flares in most patients. Studies indicate that about two-thirds of patients who stop all treatment will relapse within a year. Therefore, most clinicians maintain a low-dose DMARD long-term and only reduce biologics to the minimum needed to maintain remission.
How often for rheumatologist review
Long-term RA care requires regular monitoring, both for disease activity and drug safety. As mentioned, active RA patients should be seen monthly by rheumatology until remission/low disease activity is achieved. Thereafter, a typical schedule is every six months and should ensure all aspects of health are addressed.
The annual review ideally includes: An assessment of joint damage/ progression (clinical exam, possibly imaging); an assessment of functional ability (eg, Health Assessment Questionnaire); evaluation of any new condition — such as cardiovascular disease, which is more prevalent in RA, and osteoporosis especially if there is steroid use, depression, etc — and screening for complications of RA, such as vasculitis, pulmonary, or ocular manifestations.
RA is a systemic disease — patients may need periodic pulmonary function tests or x-rays if on certain medications or if respiratory symptoms develop (due to MTX lung or RA-associated interstitial lung disease). They may need DEXA scans if on prolonged steroids, and monitoring of cardiovascular risk factors (blood pressure, cholesterol) as chronic inflammation confers higher CV (cardiovascular) risk.
Psoriatic arthritis
PsA is a heterogeneous inflammatory arthritis associated with psoriasis, typically seronegative for rheumatoid factor. It affects about 20-to-30 per cent of people with psoriasis and can present with various patterns. It may involve the small joints of the hands/ feet (often asymmetrically), larger joints such as knees, the spine (axial PsA), and uniquely can cause ‘sausage swelling’ of an entire finger or toe and inflammation at tendon insertions, eg Achilles tendon. Early recognition of PsA is crucial because joint damage can occur if untreated, and diagnosis is challenging due to lack of a single definitive test. A high index of suspicion is needed in any psoriasis patient reporting joint pain or stiffness.
NICE Guideline NG65 (2017) on spondylarthritis, which encompasses PsA, advises that a normal rheumatoid factor, or normal inflammatory markers, for instance, do not exclude PsA. All patients with psoriasis should be assessed as per NICE, often using a simple questionnaire tool like the PEST (Psoriasis Epidemiology Screening Tool). The PEST asks about joint pain, swollen joints, nail pitting, and dactylitis — a score ?3 points warrants rheumatology referral.
Early referral to a rheumatologist is emphasised as soon as PsA is suspected – even mild joint symptoms in a psoriasis patient should be evaluated because PsA can smolder and then progress. The NICE NG65 guideline provides detailed criteria for referral – for example, any patient with a swollen digit should be
referred to rheumatology. Persistent enthesitis (tendon insertion pain) without other explanation, especially with a family history of psoriasis or presence of nail changes, should prompt referral. If inflammatory back pain is present, suggesting axial spondylarthritis, referral is indicated if certain clinical criteria are met, such as onset before the age of 45, improvement with exercise, etc. It is notable that about 15 per cent of patients develop arthritis before skin psoriasis; thus, the absence of a known history of psoriasis does not rule out PsA as they might have occult or minimal psoriasis or a family history.
What occurs after rheumatologist referral? Once referred, rheumatologists diagnose PsA through clinical examination and investigations to rule out other causes, eg, rheumatoid factor and anti-CCP are usually negative in PsA. Imaging such as x-rays or MRIs can assist, as PsA can cause characteristic changes such as joint erosions with adjacent bone proliferation — ‘erosive arthritis with new bone formation’ — and fluffy periostitis. X-ray or MRI evidence of sacroiliitis would confirm axial involvement. Given the overlap with other conditions, the diagnostic process can be complex.
Management of PsA can require many different health professionals
The management of PsA involves rheumatologists, dermatologists, primary care, and often physical therapists. Collaboration between rheumatology and dermatology is crucial. Skin and joint symptoms can fluctuate independently — for example, a patient’s psoriasis might flare while their arthritis is controlled on a TNF inhibitor that doesn’t fully cover skin disease, necessitating dermatology input for additional therapies, such as switching to an IL-17 inhibitor which
It is notable that about 15 per cent of patients develop arthritis before skin psoriasis
may better address both. Ideally, patients with severe psoriasis and PsA have co-ordinated care plans, with some centres even running combined rheum-derm clinics.
Treatment of PsA
Treatment strategies in PsA share similarities with RA in utilising DMARDs and biologics, but there are differences tailored to PsA’s clinical manifestations, such as skin involvement and enthesitis (inflammation of the entheses — the sites where tendons or ligaments insert into the bone).
For patients with milder disease or predominantly axial symptoms of pain and stiffness, NSAIDs like naproxen and diclofenac can relieve inflammatory back pain and peripheral arthritis symptoms. They may even assist enthesitis pain. As in RA, NSAIDs should be used at the lowest effective dose with monitoring for side-effects. Many PsA patients are younger (PsA onset can often be in he 30s-to-50s) but their treatment still requires caution if risk factors for NSAID toxicity exist. Simple analgesics like paracetamol can be adjuncts, and short-term may be used for severe pain or flares, though chronic use is avoided.
Oral steroids are used more sparingly in PsA than in RA, partly due to concern (largely anecdotal) that tapering off systemic steroids might trigger a psoriasis flare. However, short courses of oral prednisolone
can be used for severe flare-ups of arthritis, with careful tapering. NICE guidelines allow for short-term oral prednisolone in peripheral arthritis if NSAIDs are insufficient.
csDMARDs
For active PsA, csDMARDs — especially MTX — are the mainstay to prevent joint damage and so
are commonly used in PsA, with the added benefit of helping some patients’ skin psoriasis. MTX is typically the first csDMARD chosen, especially in patients with multiple swollen joints and is also frequently used for PsA, with evidence of benefit in peripheral arthritis.
Leflunomide in particular has shown efficacy in PsA for both arthritis and possibly skin to some degree.
It may assist with peripheral joint symptoms but is not effective for axial disease. Historically, Leflunomide has been used mainly for psoriasis, but occasionally in PsA for severe cases, although its side-effect profile limits use. Notably, it is used with caution in PsA, as some dermatologists believe hydroxychloroquine can exacerbate psoriasis skin lesions. This is a debated point, but many clinicians avoid Hydroxychloroquine (HCQ) in PsA unless absolutely needed, and if used, patients are warned to report any skin worsening.
The efficacy of csDMARDs in PsA is less predictable than in RA. For example, MTX is effective for peripheral arthritis and skin but may not adequately treat enthesitis or axial involvement (including the spine and sacroiliac joints, which are joints between spine and pelvis). Nevertheless, guidelines including NICE and EULAR recommend starting a DMARD when persistent synovitis is present in PsA. Often, a trial of two different csDMARDs is trialed before escalating to biologic therapy, unless the disease is very severe.
Biologics
PDE4 inhibitor (oral biologic)
One DMARD-equivalent to note in PsA is an oral small-molecule drug (PDE4 inhibitor) indicated for moderate PsA (and psoriasis) in adults who cannot take or fail other DMARDs. Apremilast (Otezla) is not
an immunosuppressant in the classic sense (it modulates inflammatory pathways via cyclic AMP) like traditional DMARDs. It is an attractive option for some patients who want to avoid injectable therapies and do not have severe enough disease to qualify for biologics under reimbursement criteria. However, apremilast can cause
The efficacy of csDMARDs in PsA is less predictable than in RA
significant GI side-effects (diarrhoea, nausea) and sometimes mood changes.
As with RA, MTX, leflunomide, sulfasalazine, etc, are required typically every two weeks initially, then monthly for three-to-six months, then every two-to-three months once stable. Shared care protocols in Ireland mean GPs might do this monitoring under rheumatologist guidance. PsA patients, like RA patients, should know that DMARDs may take weeks to demonstrate benefit and to keep adherence, even if symptoms are not immediately better. They should also report any side-effects or signs of toxicity — mouth ulcers for MTX, unexplained cough, etc.
PsA is commonly defined as ?3 tender and ?3 swollen joints, with failure of at least two csDMARDs, the choice of biologic can be guided by the dominant domains of disease (joints, skin, enthesitis, axial) and patient factors.
TNF inhibitors
These are often the first-line biologics in PsA as well as RA. Agents like etanercept, adalimumab, infliximab, golimumab, and certolizumab have all shown efficacy in both arthritis and skin psoriasis. They also assist with enthesitis and dactylitis (local inflammatory effects of SsA). If a PsA patient has significant spinal involvement (axial PsA like ankylosing spondylitis), TNF inhibitors are very effective for axial symptoms, whereas csDMARDs are not. Adalimumab and infliximab can also treat concomitant inflammatory bowel disease, which is relevant, as PsA (and psoriasis) have increased incidence of Crohn’s disease and ulcerative colitis. Etanercept, while good for joints, is less effective for inflammatory bowel disease, so that may influence the choice.
Interleukin-17 (IL-17) Inhibitors
Secukinumab (Cosentyx) and ixekizumab (Taltz) are monoclonal antibodies against IL-17A, a cytokine central to both psoriasis and PsA pathology. These are options typically after TNF inhibitor inadequate response, or even first-line biologic in certain scenarios — for instance, a patient with very severe psoriasis but mild arthritis might go straight to IL- 17 blocker. These drugs are given via subcutaneous injection (secukinumab monthly after a loading schedule). They are highly effective for skin lesions and also assist joints, enthesitis, and dactylitis. Side-effects include candida infections (mucocutaneous) due to IL-17’s role in mucosal immunity, and rarely inflammatory bowel disease flare (IL-17 inhibitors are avoided in patients with known IBD).
Interleukin-12 and Interleukin-23 Inhibitor (IL-12/23 Inhibitor) Ustekinumab (Stelara) targets the p40 sub-unit common to IL-12 and IL-23, cytokines involved in psoriasis. It is given as an initial injection, then every 12 weeks. Ustekinumab is effective for skin psoriasis and moderately effective for PsA joints, and it is often used if TNF inhibitors are not suitable. Newer IL-23 inhibitors (like guselkumab) are emerging with evidence in PsA, though at the time of writing, TNF and IL-17 inhibitors are more commonly used for PsA in clinical practice.
Janus kinase (JAK) inhibitor
Tofacitinib (Xeljanz) is licensed for PsA in patients who have failed biologics and, more recently, upadacitinib has also been approved for PsA. These oral agents can improve joint symptoms like enthesitis, and tofacitinib (another JAK inhibitor) has some efficacy for skin. They provide an oral alternative to injectable biologics, which some patients prefer. The safety profile and monitoring for JAK inhibitors in PsA is the same as in RA (infection risk, etc), and indeed they have similar cautions (they should not be combined with biologics, require screening, etc).
Phosphodiesterase 4 (PDE4) inhibitor
Apremilast was discussed under csDMARDs but is technically a targeted therapy for PsA. A completely different approach is, for example, if one or two joints remain inflamed, rheumatologists may inject them even if the patient is on systemic therapy, to gain additional local control.
Written by Eamonn Brady MPSI (Pharmacist ). Whelehans Pharmacies, 38 Pearse St and Clonmore, Mullingar. Tel 04493 34591 (Pearse St) or 04493 10266 (Clonmore). www.whelehans.ie. Eamonn specialises in the supply of medicines and training needs of nursing homes throughout Ireland. Email info@whelehans.ie
References
NICE Guideline NG100: Rheumatoid arthritis in adults: Management. 2018.
NICE Guideline NG65: Spondylarthritis in over 16s: Diagnosis and management. 2017. • EULAR 2019 Update: Management of rheumatoid arthritis with synthetic and biological DMARDs.
Singh JA et al. 2015 American College of Rheumatology guideline for the treatment of RA.
Smolen JS, Aletaha D, McInnes IB. Rheumatoid arthritis. Lancet. 2016;388(10055):2023-2038.
Coates LC et al. 2016 GRAPPA treatment recommendations for psoriatic arthritis.
Taylor WJ et al. CASPAR criteria for psoriatic arthritis. Arthritis Rheum. 2006.
McInnes IB, Schett G. Pathogenetic insights from the treatment of psoriatic arthritis. Lancet. 2017.
Haroon M et al. Clinical predictors of early psoriatic arthritis. Arthritis Rheum. 2014.
British Society for Rheumatology Guidelines: Prescribing biologics in rheumatology. 2022.
EMA Recommendations on JAK inhibitors (Tofacitinib, Baricitinib, Upadacitinib). 2023.
HSE High Tech Drug Scheme. Ireland: Patient access and reimbursement framework.
National Centre for Pharmacoeconomics. Evaluations of RA and PsA medications.
Pharmacy News Ireland. The pharmacist’s role in inflammatory arthritis. 2023.
Irish Statute Book: Medicinal Products (Prescription and Control of Supply) (Amendment) Regulations.
Arthritis Ireland. Pharmacist-led patient education initiatives. 2022.
Treat-to-target in psoriatic arthritis. Ann Rheum Dis. 2020; 79:685–699.
Biosimilar switching and adherence in rheumatology. BMJ 2016;352: i433.
Upadacitinib efficacy in psoriatic arthritis. PubMed ID: 32909939.
Department of Health, Ireland: Sláintecare Implementation Strategy & Action Plan 2021-2023.