Eamonn Brady MPSI provides an overview of intravenous administration of nutrients
Parenteral nutrition (PN) refers to the intravenous administration of nutrients to patients who are unable to meet their nutritional requirements via the gastrointestinal (GI) tract. Unlike oral or enteral feeding, PN bypasses the gut entirely and delivers macronutrients (amino acids, glucose, and lipid emulsions), micronutrients (electrolytes, vitamins, and trace elements), and fluid directly into the systemic circulation. It is therefore a highly specialised form of nutritional support, reserved for situations where the GI tract is either non-functional, inaccessible, or unsafe to use.
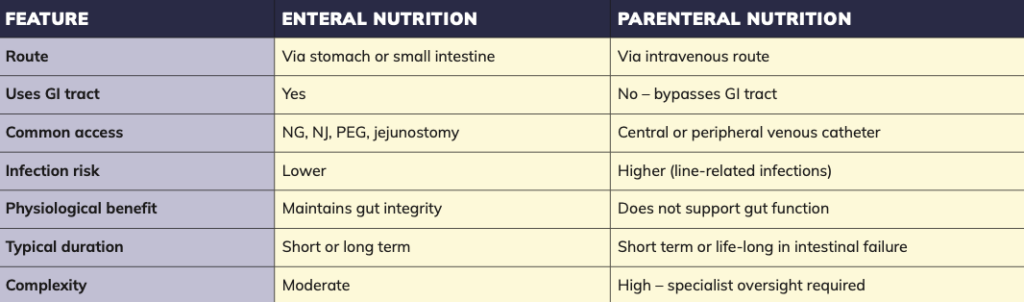
PART 1: DISTINGUISHING PARENTERAL NUTRITION FROM ENTERAL NUTRITION
Part 1a – Enteral nutrition and why is it used
Explaining enteral nutrition (via a feed- ing tube) before focusing on parenteral nutrition is important.
The fundamental principle under-pinning clinical nutrition is that if the gut works, it should be used. Enteral nutrition supports gut integrity, reduces infectious complications, and is associated with improved outcomes in many patient groups.
Enteral feeding means giving nutrition by mouth or through a feeding tube that goes into the stomach or bowel.
Why some people need tube feeding (enteral nutrition) Enteral nutrition, often called tube feeding, is used when a person cannot eat enough food by mouth, but their stomach and intestines are still working. The aim is to make sure the body gets the energy, protein, vitamins, and fluids it needs to stay healthy and recover from illness.
Below are the main reasons why someone may need to receive nutrition through a feeding tube.
1. Difficulty swallowing
Some people cannot swallow food or drinks safely. This can cause food to go into the lungs instead of the stomach, which can lead to choking or chest infections. Swallowing problems can occur after a stroke, with conditions such as Parkinson’s disease or motor neurone disease, or in advanced dementia.
2. Reduced alertness or unconsciousness
People who are very drowsy, unconscious, or sedated in hospital or intensive care cannot eat or drink safely. Tube feeding allows nutrition to be given while they recover.
3. Cancer affecting eating or swallowing
Cancers of the mouth, throat or oesophagus, or treatments such as radiotherapy, can make eating painful or impossible. Even though the gut still works, food cannot be taken by mouth.
4. Severe illness, injury, or surgery
People who are critically ill, badly injured, or recovering from major surgery often need more nutrition than they can manage to eat. Tube feeding helps support healing and recovery.
5. Poor appetite and weight loss
Some illnesses cause severe loss of appetite or rapid weight loss. Tube feeding may be used temporarily to prevent malnutrition and loss of strength.
6. Digestive conditions where liquid feeding is better tolerated In some bowel conditions, solid food may worsen symptoms, but liquid nutrition can still be absorbed. Tube feeding can help rest the gut while maintaining nutrition.
7. Feeding difficulties in babies and children
Some infants and children cannot feed normally due to prematurity, develop- mental conditions, or physical abnormalities. Tube feeding supports normal growth and development.
How enteral feeding is fitted
Healthcare professionals fit tubes. The type of tube used depends on how long the person is expected to need tube feeding.
For short-term feeding, a soft tube is passed through the nose and gently guided down into the stomach or small intestine. This is usually done at the bedside in hospital and does not require surgery. Before feeding begins, checks are carried out to make sure the tube is in the correct position.
For longer-term feeding, a tube may be placed directly through the skin into the stomach or small intestine. This is carried out as a planned medical pro- cedure using a small camera or imaging guidance. Once healed, the tube is secure and can usually be hidden under clothing.
The feed and equipment needed
Enteral feeding uses specially designed liquid nutrition that provides all the essential nutrients the body needs, including energy, protein, vitamins, minerals, and fluids.
The main equipment used includes:
- A feeding tube.
- Ready-made liquid feed in bottles or bags.
- A feeding pump or gravity system to control the rate of feeding.
- Giving sets (tubing that connects the feed to the tube).
- Syringes used to flush the tube with water before and after feeding.
- A stand or carry pack to hold the feed container.
- Feeds may be given slowly over sev- eral hours or at set times during the day, depending on the person’s needs and tolerance.
Advantages of enteral feeding
Enteral feeding has several important advantages:
It uses the digestive system, which helps keep the gut healthy.
- It lowers the risk of infection com- pared with feeding directly into a vein.
- It provides complete and balanced nutrition.
- It can be used both in hospital and at home.
- It supports recovery, wound heal- ing, and strength.
- It is a safe and effective way to provide nutrition when eating normally is not possible.
Disadvantages and potential problems
Although enteral feeding is generally safe, some disadvantages or prob- lems can occur:
- Discomfort or irritation from the tube.
- Nausea, bloating, diarrhoea, or constipation.
- Blockage or movement of the tube. fSkin irritation or infection around long-term tube sites.
- Emotional or social difficulties related to not eating normally.
Most of these issues can be man- aged with good tube care, monitoring, and support from healthcare professionals.
Short-term and long-term enteral feeding
Some people need enteral feeding only for a brief period of time. This may be due to a temporary illness
or condition, such as recovery after a stroke, major surgery, serious illness, or during cancer treatment. Once swallowing improves or normal eating becomes possible again, the feeding tube can be removed.
Other people need enteral feeding for the long term because of ongoing or permanent conditions. These may include long-term swallowing difficulties, neurological conditions, or developmental disorders. For these individuals, tube feeding provides reliable nutrition and can greatly improve health, comfort, and quality of life.
Part 1b – What is parenteral nutrition and why is it used?
Definition of parenteral nutrition
Parenteral nutrition (PN) is the intra- venous administration of nutrients, fluids, electrolytes, vitamins, and trace elements to patients who are unable to meet their nutritional requirements via the gastrointestinal (GI) tract. By definition, PN completely bypasses the gut, delivering nutrition directly into the systemic circulation. It is therefore reserved for situations in which oral or enteral nutrition is not possible, not safe, or insufficient.
PN is recognised as one of the most complex forms of nutritional support. While it can be life-saving when used appropriately, it carries a higher risk profile than enteral feeding and requires careful patient selection, close biochem- ical monitoring, and specialist multidisciplinary input.
Clinical rationale for parenteral nutrition
The fundamental principle underpinning clinical nutrition is that if the gut works, it should be used. Enteral feeding sup- ports gut mucosal integrity, preserves immune function, reduces bacterial translocation, and is associated with lower rates of infectious complications. For these reasons, enteral nutrition is always preferred where feasible.
However, there are well-established clinical scenarios in which the gastro-intestinal tract cannot be used effectively. In such circumstances, parenteral nutrition may represent the only viable option to provide adequate nutrition and prevent the consequences of starvation and severe malnutrition.
Indications for parenteral nutrition
Parenteral nutrition is indicated when enteral feeding is contraindicated or insufficient to meet nutritional requirements. Common indications include intestinal obstruction, severeileus, short
bowel syndrome, high-output enterocutaneous fistulae, severe malabsorption syndromes, and complex post-operative states following major abdominal surgery.
In selected critically ill patients, PN may be required when enteral feeding has failed or is unsafe. In oncology, PN may occasionally be used in patients with bowel obstruction or severe gastro-intestinal toxicity related to treatment, where nutritional compromise would otherwise limit ongoing therapy.
Types of parenteral nutrition
Parenteral nutrition is commonly categorised as total parenteral nutrition (TPN) or peripheral parenteral nutrition (PPN), based on the route of administration and the degree of nutritional support required.
TPN is intended to provide 100 per cent of a patient’s nutritional requirements intravenously. Due to its high osmolarity, it must be administered via a central venous catheter, usually positioned in the superior vena cava. TPN solutions are individually tailored and contain glucose, amino acids, lipid emulsions, electrolytes, vitamins, and trace elements.
PPN is administered via a peripheral vein and is limited by osmolarity constraints. As a result, it generally provides only partial nutritional support and is suitable for short-term use or as a temporary measure while central access is established.
Distinction between parenteral and enteral nutrition
It is essential to distinguish parenteral nutrition from enteral tube feeding, as the two are often incorrectly conflated. Enteral feeding involves delivering liquid nutrition directly into the stomach or small intestine via feeding tubes such as nasogastric, nasojejunal, gastrostomy, or jejunostomy tubes.
A percutaneous endoscopic gastrostomy (PEG) tube is a common method of long-term enteral feeding and does not constitute parenteral nutrition. While both approaches are used in patients who cannot eat orally, only parenteral nutrition bypasses the gastrointestinal tract entirely.
Duration of parenteral nutrition
The duration of PN varies depending on the underlying condition and the anticipated recovery of gut function. In acute hospital settings, PN may be required for days or weeks until bowel function returns or enteral feeding can be reintroduced.
A smaller cohort of patients develop chronic intestinal failure and require long-term or life-long home parenteral nutrition. These patients often have complex medical needs and require ongoing specialist support.
What is Y-site infusion?
Y-site infusion refers to the administration of two compatible intravenous medicines or solutions through a shared section of IV tubing via a Y-shaped connector, allowing them to infuse simultaneously through a single venous access point. In the context of parenteral nutrition (PN), Y-site infusion must only be used where physical and chemical compatibility has been confirmed, as in-compatibility may result in precipitation, emulsion destabilisation, or loss of drug efficacy. Careful attention to infusion rates, line-flushing, and monitoring for signs of line occlusion or adverse reactions is required to ensure patient safety.
Role of specialist services and pharmacists
In Ireland, patients requiring long-term parenteral nutrition are managed through specialist multidisciplinary nutrition and intestinal failure teams. These teams typically include physicians, dietitians, specialist nurses, microbiologists, and pharmacists.
Pharmacists play a significant role in PN management, including formulation design, aseptic compounding, compatibility and stability assessment, electro- lyte and micronutrient management, and medication review to minimise complications and drug-nutrient interactions.
PART 2: INTERACTIONS WITH PN
Part 2a – Why drug interactions with parenteral nutrition are different
Key points for pharmacists
- Parenteral nutrition should only be used when enteral feeding is not possible or insufficient.
fCentral venous access is required for TPN due to high osmolarity. - PPN is suitable only for short-term or partial nutritional support.
- Pharmacist input is essential for formulation design, stability, compatibility, and electrolyte management.
- PN carries significant risks, including infection, metabolic complications, and liver dysfunction.
- Ongoing monitoring and multidisciplinary collaboration are critical for safe PN use.
Parenteral nutrition occupies a unique position among intravenous therapies.
It is not a single drug but a complex admixture, often infused continuously over many hours through a central venous catheter. This complexity creates interaction risks that extend far beyond conventional drug-drug interactions and requires a distinct clinical approach.
PN solutions have variable pH, high osmolarity, and a dense chemical composition. Many include lipid emulsions, introducing further stability and compatibility considerations. Interactions involving PN may therefore be physical, chemical, or physiological, and they are not reliably predicted using standard interaction-checking tools.
From a practical perspective, PN-re- lated interactions can be grouped into four categories. Physical incompatibilities, such as precipitation or emulsion destabilisation, may occur when medicines come into direct contact with PN components. Chemical instability may result in degradation of either the drug or PN constituents due to pH, light exposure, or prolonged contact time. Administration-related interactions arise from Y-site co-infusion through limited venous access. Finally, clinical and metabolic interactions occur when PN composition influences glycaemic control, electrolyte balance, or fluid status, indirectly inter- acting with concurrent medicines.
These risks underpin the classification of PN as a high-risk medicine and ex- plain why pharmacists play a significant role in its governance, from prescription verification and compatibility assessment, to monitoring and education.
Part 2b – Where drug interactions occur: Mapping risk across the parenteral nutrition process
Drug interactions with parenteral nutrition rarely arise from a single point of failure. Instead, they reflect cumulative risk across the PN pathway, from prescribing and compounding, to administration and monitoring.
At the prescribing and verification stage, pharmacists must assess not only nutritional appropriateness, but also the wider medication profile
At the prescribing and verification stage, pharmacists must assess not only nutritional appropriateness, but also the wider medication profile. Limited venous access, continuous infusions, and competing administration schedules all influence interaction risk. Early identifi- cation allows alternative strategies to be explored before PN is commenced.
Compounding and formulation represent a second critical stage. PN is prepared under tightly-controlled aseptic conditions, either in hospital pharmacy units or by specialist compounding services. The addition of medicines to PN bags outside validated protocols is strongly discouraged due to unpredictable compatibility and microbiological risk. Pharmacists play a key role in reinforcing this principle and managing inappropriate requests.
The administration phase presents the most visible risk. PN is often infused over 12-to-24 hours in patients receiving multiple intravenous medicines, increasing reliance on Y-site connectors. At
this point, incompatibilities may occur rapidly, even with brief contact. Nursing practice, flushing protocols, filter use, and infusion sequencing all influence safety, making interprofessional communication essential.
Finally, monitoring and review are integral to interaction management. Altered glycaemic control, electrolyte disturbances, rising triglycerides, or abnormal liver function tests may reflect interactions between PN composition and concurrent medicines. Ongoing review allows pharmacists to anticipate problems and recommend timely adjustments.
Section 2c – Medicines that interact with parenteral nutrition
Medicines may interact with parenteral nutrition through physical, chemical, administration-related, or metabolic mechanisms, each of which carries distinct clinical implications. Understanding these interactions is essential for safe practice, so to summarise the main types of interaction:
- Physical interactions occur when medicines come into direct contact with parenteral nutrition components, leading to visible or invisible incompatibilities such as precipitation, phase separation, or emulsion destabilisation. These inter- actions can result in catheter occlusion or particulate infusion, even when no obvious visual change is observed. fChemical interactions involve chang- es in drug or nutrient stability due to factors such as pH, light exposure, or prolonged contact time. These may lead to degradation of medicines or vitamins within the PN admixture, reducing therapeutic efficacy or compromising nutritional integrity.
- Administration-related interactions arise from practical challenges associated with PN delivery, particularly Y-site co-infusion through limited venous access. Competition for intravenous access, inadequate flushing, or inappropriate sequencing of infusions can increase the risk of incompatibility or interruption of critical therapies.
- Metabolic interactions occur when the composition of PN influences the patient’s physiological response to medicines. High-glucose loads, lipid content, and electrolyte shifts may alter glycaemic control, exacerbate drug side-effects, or precipitate conditions such as refeeding syndrome, particularly in vulnerable or malnourished patients.
More details on these interactions:
Physical incompatibilities
PN solutions are complex admixtures, and direct contact with incompatible medicines may result in precipitation or instability. Calcium-phosphate precipitation is a well-recognised risk influenced by concentration, pH, temperature, amino acid composition, and contact time. Medicines containing calcium or phosphate salts, or those that alter pH, may increase this risk. Importantly, the absence of visible precipitate does not guarantee safety. Highly acidic or alkaline medicines may also precipitate PN components during Y-site administration. For this reason, PN should be considered incompatible with most medicines unless compatibility is clearly established.
Lipid emulsions
Lipid-containing PN introduces additional risks. Lipid emulsions are oil-in-water systems that may destabilise when exposed to extreme pH, divalent cations, or incompatible solvents. Destabilisation can result in emulsion cracking or coalescence, increasing embolic risk.
Compatibility data for lipid-containing PN is more limited than for aqueous solutions, and many centres avoid Y-site administration with lipid unless compatibility is explicitly documented. Appropriate in-line filtration provides an additional safeguard.
Y-site co-infusion in practice Despite best practice recommendations, Y-site co-infusion is often unavoidable in acute and critical care. A hierarchy of safety is useful: Separate lumen where possible, time separation of infusions, Y-site only when compatibility data
Antimicrobials vary widely in compatibility and may conflict
with PN schedules
exist, and documented multidisciplinary decision-making when data are lacking.
Antimicrobials vary widely in compatibility and may conflict with PN schedules. Vasoactive agents are rarely suitable for Y-site co-infusion. Electrolyte infusions, particularly potassium, magnesium, and phosphate, are frequently incompatible. Pharmacists play a key role in consulting compatibility references, advising on sequencing, and ensuring clear documentation.
Clinical and metabolic interactions
The composition of PN can significantly influence a patient’s response to medicines. High-glucose loads often necessitate insulin adjustment, particularly in patients receiving corticosteroids or catecholamines. PN may also interact with medicines, affecting electrolyte balance, increasing the risk of hypokalaemia, hypomagnesaemia, or hypo- phosphataemia.
Refeeding syndrome represents a serious metabolic interaction risk during PN initiation, particularly in malnourished patients. It occurs when the reintroduction of nutrition triggers an insulin-mediated shift of electrolytes (notably phosphate, potassium, and magnesium) into cells, leading to potentially life-threatening deficiencies. Concurrent medicines may exacerbate or mask early warning signs, requiring careful monitoring.
High-risk practices
The addition of medicines directly to PN bags outside validated protocols is almost always inappropriate. High-risk substances include concentrated electrolytes, phosphate, calcium salts, and bicarbonate. Adsorption of medicines to tubing or filters may also reduce drug delivery. When uncertainty exists, the safest option is to separate lines, separate timing, or escalate for multi- disciplinary review.
Part 2d – Preventing and managing parenteral nutrition drug interactions: The pharmacist’s role
Preventing PN-related drug inter- actions requires a proactive, systems-based approach. Early pharmacist involvement allows risks to be identified before therapy begins, including venous access limitations and competing infusions.
During administration, pharmacists should promote standardised safety hierarchies, clear guidance for nursing staff, and robust documentation when compatibility data are absent. Monitoring of glucose, electrolytes, triglycerides, and liver function tests allows early identification of metabolic interactions.
In home parenteral nutrition, the pharmacist’s role extends into patient and carer education, liaison with specialist teams, and careful review of medicine changes in the community. Even minor adjustments may have significant implications for PN safety.
Part 2e – Irish practice, governance, and key resources
In Ireland, PN delivery is guided by multidisciplinary principles promoted by IrSPEN and INDI, with pharmacists recognised as core members of nutrition support teams. UK resources from BAP- EN provide practical support, particular- ly around medicines management and transitions of care.
At European level, ESPEN guidelines reinforce cautious, evidence-based PN use and regular review. ASPEN resources offer detailed guidance on compatibility and lipid emulsions, complementing European practice.
Across all guidance, common themes emerge: PN is a high-risk medicine, compatibility must never be assumed, and uncertainty should prompt escalation. Pharmacists remain pivotal in preventing interactions, supporting patient safety, and ensuring PN delivers benefit without avoidable harm.
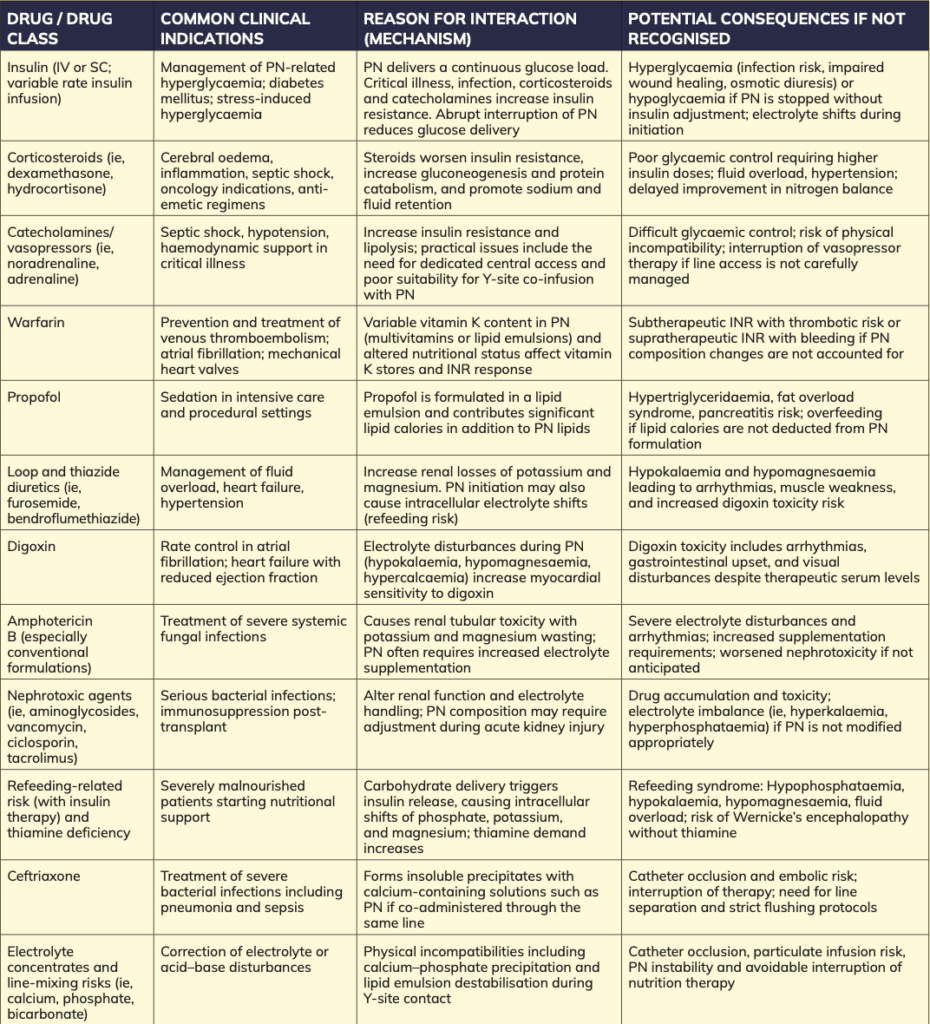
Part 2f – Specific examples of clinically relevant drug interactions with parenteral nutrition
Table 2 highlights drug interactions commonly encountered during parenteral nutrition. It includes typical indications for each drug, the mechanism of interaction with PN, and the potential clinical consequences if not recognised.
Practice notes: Compatibility data should always be checked before Y-site administration. Where uncertainty exists, use separate lumens or time-separated infusions and document decisions clearly.
Written by Eamonn Brady MPSI (Pharmacist). WhelehanAs Pharmacies, 38 Pearse St and Clonmore, Mullingar. Tel 04493 34591 (Pearse St) or 04493 10266 (Clonmore). www.whelehans.ie. Eamonn specialises in the supply of medicines and training needs of nursing homes throughout Ireland. Email info@whelehans.ie
References
• Irish Society for Clinical Nutrition and Metabolism (IrSPEN). Clinical nutrition guidelines and position statements. IrSPEN, Ireland. Updated periodically; latest accessed 2025.
• Irish Nutrition and Dietetic Institute (INDI). Parenteral nutrition practice resources
and professional guidance. INDI, Ireland, 2023–2025 editions.
• British Association for Parenteral and Enteral Nutrition (BAPEN). Introduction to parenteral nutrition. BAPEN, United Kingdom, updated 2022.
• British Association for Parenteral and Enteral Nutrition (BAPEN) and British Pharmaceutical Nutrition Group (BPNG). Medicines management and nutrition support. UK guidance documents, 2021– 2023.
• Pironi L, Arends J, Baxter J, et al. ESPEN guideline on home parenteral nutrition. Clinical Nutrition. 2020;39(6):1645–1666. • Singer P, Blaser AR, Berger MM, et al. ESPEN guideline on clinical nutrition in the intensive care unit. 2019;38(1):48–79.
• Ayers P, Adams S, Boullata J, et al. ASPEN parenteral nutrition safety consensus recommendations. Journal of Parenteral and Enteral Nutrition (JPEN). 2014;38(3):296– 333.
• American Society for Parenteral and Enteral Nutrition (ASPEN). Parenteral nutrition compatibility and stability guidelines. ASPEN, USA. Online resource, accessed 2025.
• American Society for Parenteral and Enteral Nutrition (ASPEN). Lipid injectable emulsions: safety recommendations. ASPEN, USA, 2020 update.
• National Institute for Health and Care Excellence (NICE). Nutrition support for adults: oral nutrition support, enteral tube feeding and parenteral nutrition (CG32). NICE, UK. Published 2006; last updated 2017.
• British Pharmaceutical Nutrition Group (BPNG). Medicines and nutrition support guidance for pharmacists. BPNG, UK, 2022 edition.
• Royal Pharmaceutical Society (RPS). Medicines, ethics, and practice: the professional guide for pharmacists –injectable medicines section. RPS, London, 2024.
• Mueller CM (Ed.). The ASPEN adult parenteral nutrition handbook. 3rd ed. ASPEN, USA, 2014.
• Mirtallo JM, Canada T, Johnson D, et al. Safe practices for parenteral nutrition. Journal
of Parenteral and Enteral Nutrition (JPEN). 2004;28(6):S39–S70.
• Trissel LA. Handbook on injectable drugs. 21st ed. American Society of Health-System Pharmacists, Bethesda, MD, 2023.
• White R, Bradnam V. Handbook of drug administration via enteral feeding tubes. 3rd ed. Pharmaceutical Press, London, 2015.
• Crook MA, Hally V, Panteli JV. The importance of the refeeding syndrome. Nutrition. 2001;17(7–8):632–637.
• Boateng AA, Sriram K, Meguid MM, Crook M. Refeeding syndrome: treatment considerations based on collective analysis of literature case reports. Nutrition. 2010;26(2):156–167.
• National Patient Safety Agency (NPSA). Reducing harm from intravenous medicines. Patient Safety Alert. NPSA, UK, 2007.
• Institute for Safe Medication Practices (ISMP). Parenteral nutrition safety alerts and best practice recommendations. ISMP, USA. Ongoing updates, accessed 2025.
• European Medicines Agency (EMA). Quality and safety considerations for parenteral nutrition products. EMA, European Union. Regulatory guidance, accessed 2025.
• Health Service Executive (HSE). National nutrition and hydration policy and supporting frameworks. HSE, Ireland, 2018–2024 updates.
• Boullata JI, Gilbert K, Sacks G, et al. A.S.P.E.N. clinical guidelines: parenteral nutrition ordering, order review, compounding, labelling, and dispensing. Journal of Parenteral and Enteral Nutrition (JPEN). 2014;38(3):334– 377.
• Worthington P, Balint J, Bechtold M, et al. When is parenteral nutrition appropriate? JPEN. 2017;41(3):324–377.
• Parrish CR, Krenitsky J. Refeeding syndrome: practical guidelines for prevention and treatment.
• Practical Gastroenterology. 2016;40(9):32– 44.
• Newton DW. Drug incompatibility chemistry. American Journal of Health-System Pharmacy. 2009;66(4):348–357.
• McClave SA, Taylor BE, Martindale RG, et al. Guidelines for the provision and assessment of nutrition support therapy in the adult critically ill patient. JPEN. 2016;40(2):159–211.
• Mirtallo JM, Dasta JF, Kleinschmidt KC, Varon J. State of the art review: intravenous fat emulsions.
• Pharmacotherapy. 2010;30(9):1006–1024. • Driscoll DF. Lipid injectable emulsions: pharmacopeial and safety considerations. Nutrition in Clinical Practice. 2017;32(1):96– 107.
• Royal Pharmaceutical Society (RPS). Professional guidance on the administration of injectable medicines. RPS, London. Latest edition 2023–2024.
• Institute for Safe Medication Practices (ISMP). High-alert medications in acute
care settings – parenteral nutrition. ISMP Medication Safety Alert. Updated regularly; accessed 2025.
• British Pharmaceutical Nutrition Group (BPNG). Parenteral nutrition: a practical guide for pharmacists.
• Pharmaceutical Journal. 2020;305(7938). • Boullata JI, Gilbert K, Sacks G, et al. A.S.P.E.N. clinical guidelines: parenteral nutrition ordering, order review, compounding, labelling, and dispensing. Journal of Parenteral and Enteral Nutrition (JPEN). 2014;38(3):334–377.
• Mirtallo JM, Canada T, Johnson D, et al. Safe practices for parenteral nutrition. Journal of Parenteral and Enteral Nutrition (JPEN). 2004;28(6):S39–S70.
• Newton DW. Drug incompatibility chemistry. American Journal of Health- System Pharmacy. 2009;66(4):348–357.
• Trissel LA. Handbook on Injectable Drugs. 21st ed. American Society of Health-System Pharmacists, Bethesda, MD. 2023.
• Ayers P, Adams S, Boullata J, et al. ASPEN parenteral nutrition safety consensus recommendations. Journal of Parenteral and Enteral Nutrition (JPEN). 2014;38(3):296– 333.
• Institute for Safe Medication Practices (ISMP). High-alert medications in acute
care settings: parenteral nutrition. ISMP Medication Safety Alert. Updated regularly; accessed 2025.