Dr Donna Cosgrove MPSI synopsises kidney disorders, including the most common risk factors
Introduction
Chronic kidney disease (CKD), also known as chronic renal failure, is defined as abnormalities of kidney structure or function that are present for ?three months and have negative implications for health. The most common cause is diabetes, with approximately 20-to-40 per cent of people with diabetes developing kidney disease. Hypertension is the second-most common cause.
Other risk factors include age (>50 years), obesity, male sex, Black or Hispanic ethnicity, family history, smoking, long- term analgesic use, and autoimmune disorders. Monogenic disorders account for up to 30 per cent of non-diabetic CKD in adults, most commonly due to PKD1 and PKD2 gene variants.1
CKD prevalence is approximately 11 per cent in high-income countries and is more prevalent in individuals with lower socioeconomic backgrounds. Those in the lowest socioeconomic quartile have a 60 per cent higher risk of progressive CKD than those in the highest.
While socioeconomic status contributes to this disparity, it does not fully explain the increased risk seen in certain ethnic groups. For example, Black and Asian populations in the UK have higher CKD risk.2 Symptoms of CKD are often non-specific, such as fatigue and nausea, progressing to oedema, pruritus, dry skin, and anorexia in later stages. If kidney dysfunction is suspected, blood tests assessing renal chemistry (eg, electrolytes, urea, creatinine, glucose) and glomerular filtration rate (GFR) should be performed. Optimising glycaemic control and blood pressure is essential to slow disease progression.1
Pathophysiology
Kidney damage initiates a cascade of adaptive and maladaptive processes;
- Increased intraglomerular pressure occurs as the kidney attempts to maintain its filtration function despite nephron loss, resulting in glomerular hypertrophy.
- Glomerular permeability increases, allowing macromolecules such as inflammatory mediators and proteins to accumulate, contributing to cellular toxicity, inflammation, fibrosis, and scarring.
- Upregulation of angiotensin II promotes transforming growth factor beta (TGF-?) activity, driving collagen deposition, leading to stiffening and further glomerular damage.
- Tubulointerstitial disease is also present in all forms of CKD. Although mechanisms are not fully understood, reduced blood supply and inflammatory infiltration contribute to interstitial fibrosis and tubular atrophy, accelerating progression.
- Diagnosis of kidney disease
- GFR is the best indicator of kidney function, representing the volume of fluid filtered by
functioning nephrons over time. CKD is generally diagnosed when GFR is <60mL/ min/1.73m2, or when markers of kidney damage are present for at least three months, regardless of cause.2 Estimated GFR (eGFR) should be interpreted cautiously in patients with extremes of muscle mass. Patients should avoid meat consumption for 12 hours prior to testing.3 Markers of kidney damage include:
- Albuminuria (urine albumin-creatinine ratio [ACR] ?30mg/g);
- Urinary sediment abnormalities;
- Electrolyte abnormalities due to tubular disorders;
- Histological abnormalities;
- Structural abnormalities detected by imaging; and
- History of kidney transplantation. End-stage kidney disease (ESKD),
where kidney function cannot sustain life long-term, is indicated by GFR <15mL/min/1.73m2, requiring dialysis, transplantation, or conservative care. ACR is preferred over reagent strips for detecting proteinuria, although strips may identify haematuria. GFR should be monitored at least annually in patients taking nephrotoxic medicines (eg, calcineurin inhibitors, lithium, NSAIDs), and in individuals with risk factors including diabetes, hypertension, previous AKI, cardiovascular disease (CVD), structural renal abnormalities, recurrent kidney stones, prostatic hypertrophy, multisystem diseases, gout, family history of renal disease, or incidental haematuria/proteinuria.
CKD classification should use combined GFR and ACR categories, as outlined in Table 1.
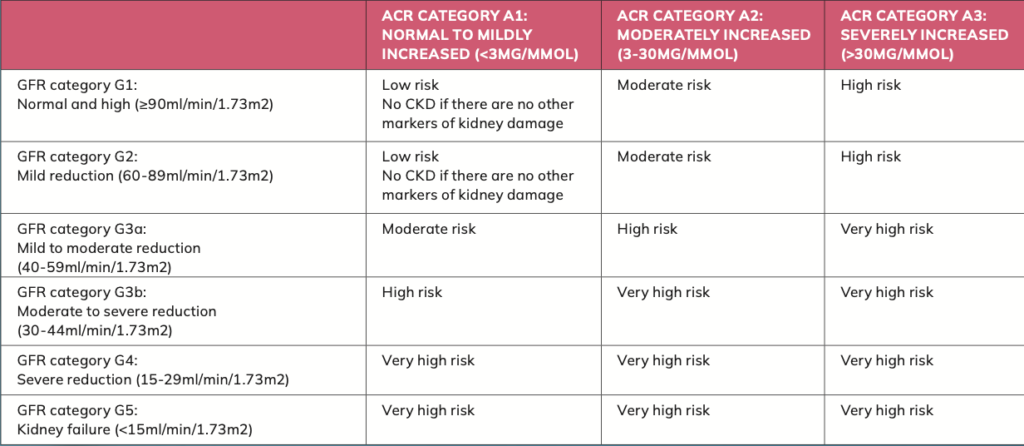
Abbreviations: ACR: albumin creatinine ratio; GFR: glomerular filtration rate
A patient-specific plan should be agreed to determine CKD cause, particularly where reversible factors exist (eg, obstruction, nephrotoxic medicines).3 Monitoring frequency should reflect CKD stage, progression rate, comorbidities, and treatment changes — ranging from annually in low-risk patients to ? four times yearly in very high-risk groups.
Lifestyle advice is essential — patients should be encouraged to exercise, maintain a healthy weight,
and stop smoking. Dietary advice may include guidance on potassium, phosphate, calorie, and salt intake where appropriate. Routine low-protein diets are not recommended.
Complications of chronic kidney disease
CKD is frequently asymptomatic until advanced stages and is often detected incidentally.2 As renal function declines, uraemic toxins accumulate, leading to systemic effects such as cognitive changes, gastrointestinal symptoms, altered urine output, oedema, pruritus, muscle cramps, dyspnoea, hypertension, and pallor due to anaemia.
Anaemia of CKD is typically normocytic and normochromic, resulting from reduced erythropoietin production. Management includes iron supplementation and erythropoiesis- stimulating agents such as epoetin alfa, epoetin beta, or darbepoetin alfa, which reduce transfusion requirements. CKD also disrupts calcium and phosphate homeostasis due to impaired vitamin D activation, leading to hypocalcaemia and secondary hyperparathyroidism, which increases bone resorption.
Cardiovascular risk is significantly elevated. Individuals with GFR <60 have approximately a 57 per cent higher risk of CVD, with increased rates of myocardial infarction and stroke as kidney function declines. Blood pressure control is therefore critical.
Cancer incidence is also increased in end-stage disease, particularly renal and thyroid cancers. This is likely multifactorial, involving immunosuppression, chronic uraemia, and underlying renal pathology.
Pharmacotherapy
Pharmacological management aims to slow CKD progression and reduce complications. Renin–angiotensin system (RAS) blockade with ACE inhibitors or angiotensin receptor blockers (ARBs)
is a cornerstone of therapy, reducing intraglomerular pressure and proteinuria. Sodium-glucose co-transporter-2 (SGLT2) inhibitors (eg, empagliflozin, canagliflozin, dapagliflozin) also preserve kidney function independently of blood pressure and glycaemic control.1
NICE guidelines recommend ACE inhibitors or ARBs as first-line treatment in patients with CKD. In patients who are over 55 years or of black African or African-Caribbean origin, calcium channel blockers are recommended initially to treat hypertension, with thiazide diuretics as alternatives if not tolerated.3 In CKD, ACE inhibitors or ARBs should be titrated to the highest tolerated dose. Blood pressure targets should be individualised based on patient factors.
In the US, the National Kidney Foundation’s ‘Kidney Disease: Improving Global Outcomes’ (KDIGO) guidelines suggest a systolic BP target <120mmHg if tolerated, while NICE recommends <140/90mmHg, or <130/80mmHg in patients with significant albuminuria.
Patients in G1-G2 stages should receive lifestyle advice alongside pharmacotherapy. SGLT2 inhibitors are recommended in patients with eGFR ?20mL/min/1.73m2. Statins should also be considered due to cardioprotective effects, with evidence showing reduced mortality and cardiovascular events in CKD patients not requiring dialysis.
Cancer incidence is also increased in end-stage disease, particularly renal and thyroid cancers
Patients with G3-G4 CKD should be counselled on renal replacement therapies, including haemodialysis, peritoneal dialysis, and transplantation. Glycaemic targets should be individualised, typically ranging from HbA1c <6.5 per cent to <8 per cent in patients not receiving dialysis.
Vitamin D deficiency should be corrected where present, and monitoring for secondary hyperparathyroidism is recommended in patients with G3a-G3b CKD.
Kidney stones
Kidney stones are mineral deposits that form in the renal calyces and urinary tract.4 They develop when urinary mineral concentrations are sufficiently high to form crystals that are retained in the kidney. Approximately 80 per cent are composed of calcium oxalate or calcium phosphate, with the remainder consisting of uric acid, struvite, cystine, or — less commonly — drug-related compounds. Stone size ranges from microscopic to several centimetres. Their presence increases the risk of CKD, end-stage renal disease, and hypertension.
Patients typically present with renal colic, characterised by severe, cramping flank pain, often accompanied by haematuria, nausea, vomiting, and occasionally fever, although some cases are asymptomatic. The global prevalence ranges from 1.7-14.8 per cent and is increasing due to lifestyle and dietary factors. Risk factors include obesity, diabetes, hypertension, metabolic disorders, and dehydration.
Management includes pain relief, facilitation of stone passage, and prevention of recurrence, with NSAIDs the recommended first-line treatment for pain. Antispasmodics are not recommended due to apparent lack of efficacy. Alpha blockers and calcium channel blockers may aid stone passage by relaxing the ureter, although specific guidance on their use and regimen is variable.5 Uric acid stones may be dissolved using urinary alkalinisation with potassium citrate. In severe cases, interventional options include shockwave lithotripsy, percutaneous nephrolithotomy, and retrograde intrarenal surgery using a ureteroscope.
Role of the pharmacist
Pharmacists can play a key role in identifying at-risk patients, optimising medicines, and preventing harm. This includes reviewing nephrotoxic medicines (eg, NSAIDs), supporting adherence, providing lifestyle advice, and recognising when referral is required.
References
1. Singh M and Krause M. (2026, March 6). Chron- ic Kidney Disease. BMJ Best Practice. Retrieved April 6, 2026, from https://bestpractice.bmj.com/ topics/en-gb/84.
2. Kalantar-Zadeh K, Jafar TH, Nitsch D, Neuen BL, and Perkovic V. (2021). Chronic kidney disease. The Lancet, 398(10302), 786-802.
3. National Institute for Health and Care Excellence. (2021). Chronic kidney disease: Assessment and management (NG203). Retrieved April 7, 2026, from www.nice.org.uk/guidance/ng203/resources/ chronic-kidney-disease-assessment-and-manage- ment-pdf-66143713055173.
4. Khan SR, Pearle MS, Robertson WG, Gambaro G, Canales BK, Doizi S … and Tiselius HG. (2016). Kidney stones. Nature Reviews Disease Primers, 2(1), 16008.
5. National Guideline Centre, UK (2019). Medical ex- pulsive therapy: Renal and ureteric stones ? assess- ment and management. Retrieved April 7 2026, from https://pubmed.ncbi.nlm.nih.gov/35133754/.