Terry Maguire looks at some pilot projects that might lead the way to a more client-centered approach to managing codeine misuse/abuse
Misuse, overuse and abuse of codeine-containing analgesics is well recognised and is becoming, if not already, a serious public health concern. Codeine, a relatively weak opioid, in combination with either paracetamol or ibuprofen, remains available over-the-counter (OTC), in my opinion against the odds. OTC, it is licensed for short-term (three days) treatment of acute, moderate pain that is unresponsive to paracetamol, ibuprofen or aspirin alone. I do wonder who uses aspirin as a painkiller these days.
There is, which will come as a surprise to most pharmacists, little evidence for the clinical benefit of low-dose codeine in combination with other analgesics.
It seems products are designed, not on evidence of efficacy to manage pain, but on the highest dose of codeine that can be sold legally OTC. Co-codamol is not more effective than paracetamol alone, according to the Cochrane Collaborative, and where the combination does not reduce pain more compared to paracetamol, it will cause constipation and can lead to dependency.
Habitual users of codeine will, of course, claim its superior pain-killing effectiveness, when they are really referring to its narcotic effect and the most frequent users are likely addicted, risking withdrawal should they stop. One of the main withdrawal symptoms is medication headache which, to their logic, requires more medicine. It’s a vicious cycle.
The true extent of the problem is difficult to quantify due to the often- hidden nature of misuse/dependency and the widespread availability of codeine products from pharmacies, including online providers. In 2023, the UK was estimated to have the third-largest consumption of codeine at over 28 tonnes after India and Italy (75 tonnes and 33.5 tonnes, respectively). The science would suggest OTC codeine be done away with, but we seem committed to trying anything short of taking this nuclear option. A number of investigations into OTC codeine use/abuse have considered options but they are largely ineffective in controlling use.
ROI has taken a strong professional regulatory approach towards pharmacists supplying OTC codeine.
In 2022, the Health Service Executive (HSE) issued a National Patient Safety Alert (NPSA), stating that: “Regular or prolonged use of codeine-containing products may produce psychological and physical dependence. For combination products, use of higher doses and/or for a longer duration than that recommended, can also lead to serious adverse clinical outcomes arising from exposure to the analgesic component (eg. paracetamol or ibuprofen). These include hepatotoxicity, gastrointestinal and renal toxicities, such as gastrointestinal haemorrhage and perforation and renal failure.” (HSE, 2022).
There is less obvious regulatory oversight on pharmacists in N Ireland or across the rest of the UK. Discussions in the UK will continue about the legal status of codeine-containing products. Some argue that the reclassification to POM will increase strain on GPs, and so urging the need for careful consultation. Nonetheless, codeine addiction will continue to be a concern regardless of the legal status and community pharmacists should have role to play in helping to address the problem, certainly if they want to retain the OTC medicine category.
The problems with codeine misuse extend beyond the UK and Ireland and regulation varies between countries. Australia, Austria and Italy have all restricted codeine supply to POM status. A recent study into the impact of rescheduling of codeine in Australia found that there were no unintended consequences and it appears to have reduced codeine use and associated harms without substitution to other opioids or overuse of alternative non- opioid analgesics. However, increased GP consultations were observed.
Non-medical use (abuse/misuse) of codeine has been reported to be between four and eight times higher in countries where it is available to purchase OTC when compared to countries where it
is restricted to POM use. A qualitative study involving the UK, Ireland and South
Africa found that pharmacists from all three countries viewed codeine misuse as a problem, however, there were mixed opinions about re-scheduling. Pharmacists in the UK and Ireland have indicated a desire to demonstrate that they can manage the problem of misuse. With significant sales in this OTC category, the commercial implications of a switch to POM would impact significantly on pharmacy businesses.
So, can pharmacists really manage this category sufficiently to reduce risk to patients? What interventions and training might be needed to allow this to happen?
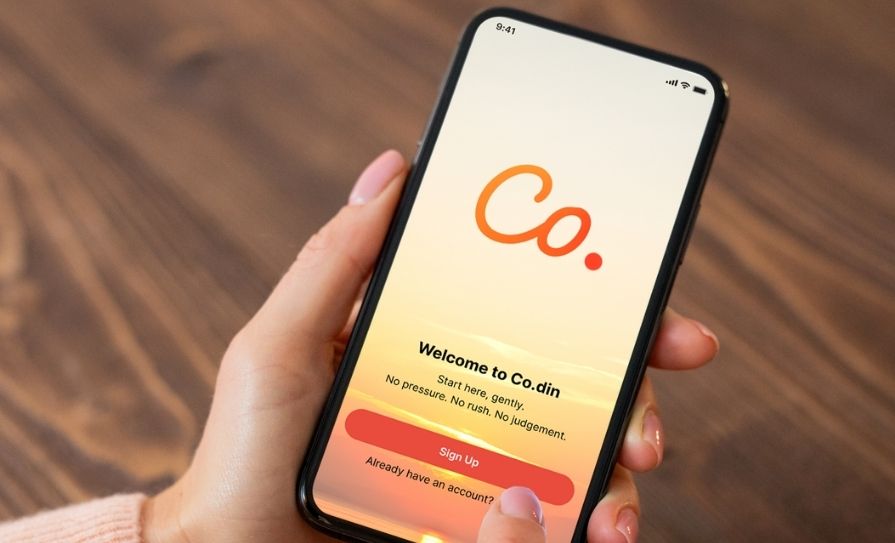
A number of interventions have been proposed. A brief four-item Codeine Dependence Scale (CDS) was used to support routine screening in the pharmacy, with some benefits noted. Currently, two interventions are being tested in N Ireland; the Co.din App, and the Health Board pharmacy intervention service.
The Co.din app
A mobile phone app, Co.din has been developed to help support codeine users identify and address problems associated with codeine use, including misuse. Co.din is a free, anonymous wellbeing app that helps people reflect on their codeine use, build healthier routines, and track small wins. The app does not provide direct individual medical advice but it does encourage users to seek medical help where they have concerns about their use of codeine, including addiction.
The Co.din App was created by Belfast Pharmacist Richard Addy and commissioned by Northern Pharmacies Trust, a charity focused on development of community pharmacy practice. The app, now ready for evaluation, will be assessed for three months in 10-to-15 pharmacies.
A principle of the App is that it, in itself, is a non-judgmental brief intervention for those potentially overusing or abusing codeine-containing analgesics and some who might not even appreciate that
they are doing so. It is designed to make clients think about what they are doing, see that this is not good for their health and wellbeing, and encourage them to take small steps to change behaviour.
The app evaluation involves a leaflet being placed in the bag with every purchase of a codeine-containing analgesic. The pharmacy team is not directly involved in challenging the behaviour.
One of four things will happen based on Prochaska and DiClemente’s Cycle of Change:
(1) The leaflet will be ignored and discarded (unwilling to change).
(2) It will be read but unactioned (thinking about change).
(3) It will be actioned confidentially (planning to change).
(4) It will be actioned and perhaps stimulate a conversation with the pharmacist (changing).
Training of pharmacy staff will take place at an evening event before the evaluation launch. This training will address brief interventions within Motivational Interviewing (MI). Brief intervention/MI is helping people think about and make change in a personal and resilient way, encouraging them to take responsibility for their own health and wellbeing and monitor their own progress while taking responsibility for that change. This is a skill set many pharmacists have difficulty developing. The less we say the better, which for most of us is counter-intuitive.
East Antrim pilot
The Health Board (SPPG) in conjunction with Public Health Agency in N Ireland are running a pilot intervention to address OTC codeine use/abuse starting in April 2026 in East Antrim pharmacies. This scheme asks pharmacists to actively engage with those purchasing OTC codeine using a brief intervention approach. That engagement will require completion of an assessment questionnaire. It is hoped that willing clients will be referred to Community Addiction Teams for assessment and where necessary, treatment.
Is there a best approach?
It is unlikely that each person encountered in a pharmacy and regularly requesting an OTC codeine analgesic will be the same.
Some will be unwilling to change, others will be keen to change, and most will be somewhere in between. Pharmacists need the skills to know which is which and this is why the training in delivering Brief Interventions and Motivational Interviewing will be critical to both projects’ success.
The objective of any brief intervention is to allow clients to get an insight to their behaviour, to appreciate that this behaviour is potentially dangerous and damaging to their health and wellbeing, and to take effective action to change the behaviour.
The East Antrim pilot should prove more successful with clients who are planning to change or in the process of change. The Co.din App may be more appropriate for those who are unwilling to, or not thinking about, change.
In the East Antrim Pilot clients, those unwilling to change may become resistant when approached in the pharmacy and this resistance could lead to more entrenched behaviours and more unwillingness to change. Rolling with Resistance is a key skill in Motivational Interviewing and pharmacists undertaking the interventions will need to develop skills that avoid argumentation, often the hallmark of current interventions.
The outcome of these pilots will be very interesting and might lead the way to a more client-centered approach to managing codeine misuse/abuse generally. Will they offer a means by which community pharmacy can retain this important medicine category, or will they be ineffective and identify the need to scrap the category and make codeine, in all its strengths, a POM?
Terry Maguire owns two pharmacies in Belfast. He is an honorary senior lecturer at the School of Pharmacy, Queen’s University Belfast. His research interests include the contribution of community pharmacy to improving public health.